The U.S. Food and Drug Administration (FDA) approved the macular degeneration medication Beovu in 2019 but criticism about Boveau’s side effects has circulated ever since.
What Is Beovu?
Beovu is an injectable medication used in the treatment of wet age-related macular degeneration (AMD). Macular degeneration is an issue with the eye’s retina – specifically the part of the retina known as the macula. The macula is responsible for central, detailed vision, so this condition can lead to the loss of central vision and fine details, while peripheral vision remains normal.
Macular degeneration can be “dry” or “wet.” Dry AMD is far more common, comprising about 80% of cases. With dry AMD, parts of the macula thin with age and grow clumps, or protein, which interfere with vision slowly.
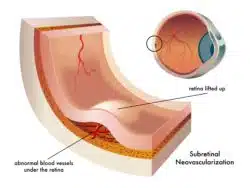
In contrast, wet AMD is caused by abnormal blood vessels that grow behind the retina. These blood vessels can leak blood and other fluids which scar the macula, causing rapid vision loss. Because vision loss occurs so rapidly with wet AMD, this condition is very serious and may require prompt treatment with medications such as Beovu.
Beovu inhibits the human vascular endothelial growth factor in the body which can cause irregular blood vessels to form – including those seen in wet AMD. Although this helps slow the progress of wet AMD, and may even improve vision, Beovu treatment may be associated with concerning side effects.
 Side Effects of Beovu Treatment
Side Effects of Beovu Treatment
Like every drug, Beovu has side effects. Common mild side effects may include:
- Blurry vision
- Cataracts
- Broken blood vessels in the eye
- Eye “floaters” which look like spots or strings in vision
- Eye pain
- Eye swelling
- Conjunctivitis (pink eye)
- Vision changes
Although these side effects may only be temporary, more serious complications that may be associated with Beovu treatment include:
- Serious eye issues such as retinal vasculitis
- Retinal vascular (vein) occlusion
- Vision loss, blindness, and other vision problems
Beovu Approval
The FDA approved Beovu in October 2019, but its safety and potential for catastrophic side effects have dogged the drug since the beginning.
Following 14 reports of vasculitis in Beovu patients, the American Society of Retina Specialists (ASRS) shared a members-only update warning its members about the issue. Retinal vasculitis is an inflammation of the blood vessels in the eye which can affect vision and even lead to vision loss.
Beovu manufacturer Novartis maintained that the drug was both safe and effective. However, the company did implement its own safety review due to the retinal vasculitis reports.
“Novartis stands behind the safety and efficacy of Beovu. In addition to our own internal assessment, we have engaged an external safety review committee (SRC) to further evaluate these post-marketing cases,” the company said in a statement.
Despite these assurances, this summer saw new safety challenges and labeling changes for the drug in order to warn the public of problems associated with Beovu treatment.
Novartis reportedly used an external safety review committee to investigate the reports of retinal vasculitis and retinal vascular occlusion. Upon reviewing events reported during drug trials, Beovu’s safety committee found that the issues could be a side effect of the treatment and occurred most commonly in the presence of intraocular inflammation.
An analysis of post-marketing data showed that retinal vasculitis, retinal vascular occlusion, or both events together occurred in about seven of every 10,000 injections. Retinal vasculitis and retinal vascular occlusion reportedly occurred in about two per 10,000 injections. Together, these events occurred in nearly four of every 10,000 injections.
Due to these risks, Novartis recommended that doctors check for intraocular inflammation before injecting patients with Beovu. However, Beovu carries its own risk for intraocular inflammation, reportedly occurring in 4% of cases.
Shortly after this announcement, the FDA approved a label update for Beovu which includes warnings about retinal vasculitis and retinal vascular occlusion.
“This label update provides clinicians with important information to guide treatment decisions. We believe Beovu continues to represent an important treatment option for patients with wet AMD, with an overall favorable benefit-risk profile,” Novartis’ global head of medical affairs and chief medical officer Marcia Kayath said in the news release.
Although Beovu’s new label accounts for some adverse side effects, patients treated in the past may not have been fully informed of the risks associated with Beovu treatment. If you experienced severe vision side effects as a result of Beovu, you may be able to file a Novartis Beovu lawsuit.
 Why Were Risks Discovered After FDA’s Beovu Approval?
Why Were Risks Discovered After FDA’s Beovu Approval?
Patients who used the drug may wonder why the risk of severe vision loss was not discovered until after Beovu approval was issued by the FDA. It is likely, according to an expert that talked to Healio News, that the studies conducted on the drug, called clinical trials, did not include measures that considered the pathology in the peripheral retina.
Indeed, there are a number of ways manufacturers can miss risk factors in drugs and medical devices and still get FDA approval. Rarely, say experts who talked to Healio News, drug and medical device makers are plagued by just plain old bad luck. Usually, one or more of the following factors results in a drug or device hitting the market without at least the severe risks known:
- There was not enough funding for the clinical trials.
- The clinical trials were not designed well.
- Research did not identify and explore the right risks.
- There was a commitment bias at play.
- The manufacturer hid results of the research trial during the approval process.
- There was a bias in play at the FDA during the approval process.
FDA approval of Beovu came after 1,800 patients took part in clinical trials for 96 months, per Healio News. Experts reportedly expected a quick adoption of the drug at the time of approval, but less than a year later, it was evident that patients were experiencing severe side effects not revealed by the clinical trials.
While drugmaker Novartis is reportedly funding additional investigation into the risks of Beovu through a Safety Review Committee, the drug remains a secondary option to safer options that were already on the market.
If you or someone you love has suffered from vision loss or other eye issues after using Beovu, you may be able to file a lawsuit and pursue compensation.
Filing a lawsuit can be a daunting prospect, so Top Class Actions has laid the groundwork for you by connecting you with an experienced attorney. Consulting an attorney can help you determine if you have a claim, navigate the complexities of litigation, and maximize your potential compensation.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.












