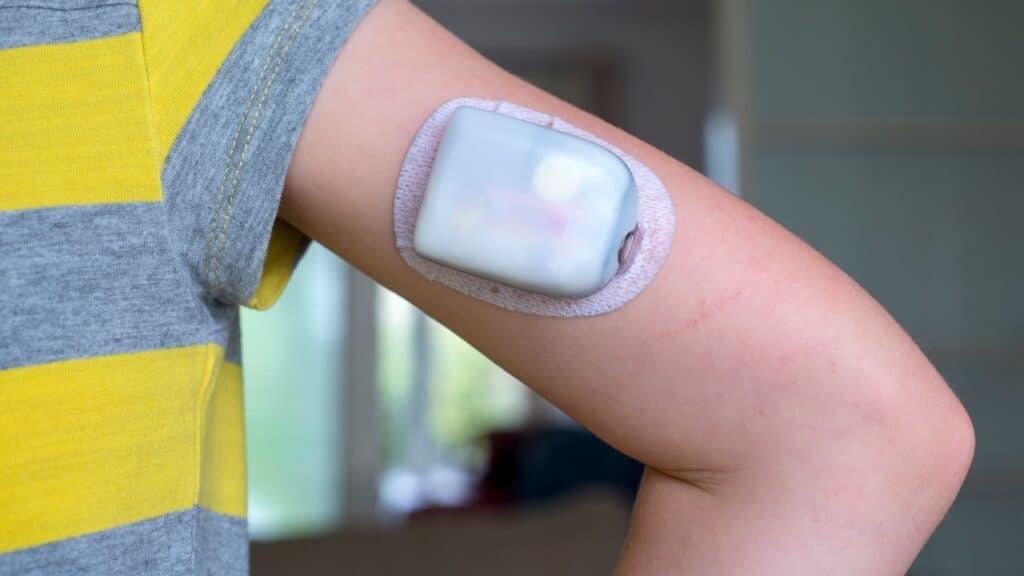
Omnipod recall overview:
- Who: Insulet Corporation is recalling specific lots of Omnipod 5 Pods, impacting approximately 1.5% of their annual production.
- Why: The recall is due to a potential tear in the internal tubing of the Pods, which may cause insulin to leak inside rather than being delivered to the body.
- Where: The recall is effective in the United States.
Insulet Corporation is recalling certain lots of its Omnipod 5 Pods due to a manufacturing defect that could lead to insulin delivery issues.
The Omnipod recall affects specific lots distributed within the United States, with the potential for insulin leakage inside the Pods, posing a risk of under-delivery to users.
The recall, announced on March 12, involves the Omnipod 5 Pods, an alternate controller-enabled insulin infusion pump. The defect identified involves a small tear in the internal tubing, which could cause insulin to leak inside the Pod rather than being infused into the body. This issue was discovered through Insulet’s ongoing product monitoring efforts.
“If insulin is not delivered properly, users may experience high blood glucose levels due to under-delivery of insulin,” the Omnipod recall says.
Insulet has reported 18 serious adverse events, including hospitalizations and diabetic ketoacidosis (DKA), linked to this defect, though no fatalities have been reported.
Insulet urges consumers to discontinue use of affected Pods
The recall notice emphasizes the importance of discontinuing the use of affected Pods and replacing them with unaffected ones.
“Customers should visit omnipod.com/check-pods to confirm whether their Pod lot number is included in this voluntary Medical Device Correction,” the recall notice says.
Insulet has assured that the issue does not impact continuous glucose monitoring systems or their readings. The company has taken steps to enhance its manufacturing processes and quality controls to prevent future occurrences of this Omnipod recall issue.
Insulet will continue to produce and distribute Omnipod 5 Pods and does not foresee any disruption in supply or availability for new patients.
For more information, consumers can visit omnipod.com/check-pods to verify if their Pods are affected and request replacements at no cost. Additionally, Insulet Product Support is available 24/7 at 1-800-641-2049 for any questions or assistance.
Insulet says it has received 18 reports of serious adverse events related to the recall so far, including hospitalizations and diabetic ketoacidosis. The company is not currently facing legal action over the recall, but Top Class Actions follows recalls closely as they sometimes lead to class action lawsuits.
Are you affected by this Insulet recall? Let us know in the comments.
Don’t Miss Out!
Check out our list of Class Action Lawsuits and Class Action Settlements you may qualify to join!
Read About More Class Action Lawsuits & Class Action Settlements:















9 thoughts onInsulet announces recall of Omnipod 5 Pods due to insulin delivery issue
Add
Yes my child was affected by this!
Add me
I have this as well as the insulin pump.
Please add me
I was affected by this, I thought it was something I was doing wrong. I also went through all of them before finding out. Im out 100$ copay plus the cost of wasted insulin plus the days I was sick and found it hard to work.
My husband ended up in the ER with ketoacidosis. It was determined that the Omnipod was defective in delivering insulin.
My husband has many problems out his last batch the tube would stop up and not give him insulin and been many times i could smell insulin but he could not get his sugar down
Yes, always had issues with the delivery of omipod.
Add me