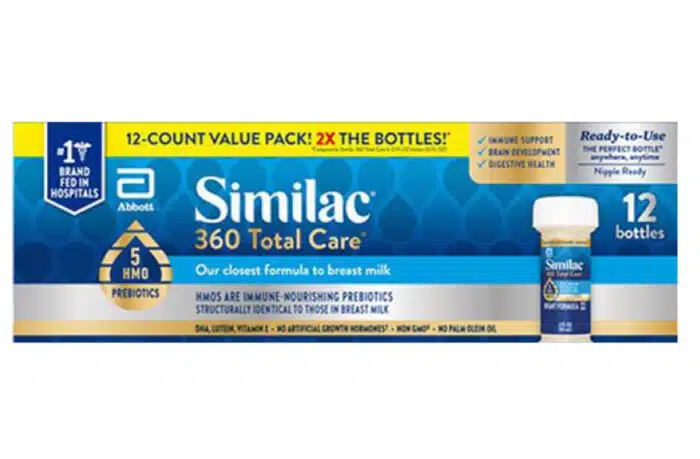
Similac, Pedialyte recall overview:
- Who: Abbott recalled certain lots of ready-to-feed liquid products for infants and children sold under various brands including Similac and Pedialyte.
- Why: Some of the bottles have caps that may not have sealed completely, which could result in spoilage.
- Where: The recall is active in the United States and 13 other countries.
Abbott recalled certain lots of ready-to-feed liquid products for infants and children sold under various brands including Similac and Pedialyte, as some of the bottles may not be sealed properly.
The U.S Food & Drug Administration (FDA) website issued the recall on Oct. 14.
It applies to certain lots of 2 fluid ounce/59 milliliter bottles of ready-to-feed liquid products for infants and children, including the brands Similac® Pro-Total ComfortTM, Similac® 360 Total Care®, Similac 360 Total Care Sensitive, Similac® Special Care® 24, Similac Stage 1, Similac® NeoSure®, Similac Water (Sterilized) and Pedialyte Electrolyte Solution.
“These products are being recalled because a small percentage of bottles (less than 1%) in the recalled lots have bottle caps that may not have sealed completely, which could result in spoilage,” the Similac and Pedialyte recall notice says. “If spoiled product is consumed, gastrointestinal symptoms such as diarrhea and vomiting may occur.”
To identify if your product is included in the recall, visit similacrecall.com to view a list of impacted lot numbers.
Pedialyte, Similac recall should not affect US infant formula supply, Abbott says
The Pedialyte and Similac recall comes after the FDA required Abbott to shut down its Sturgis facility after Similac brand baby formula produced at the plant was linked to a contamination of cronobacter sakazakii and salmonella newport that sickened several infants and led to two deaths.
The nationwide baby formula shortage forced the United States to import millions of pounds of baby formula into the country when Abbott’s Sturgis factory shuttered.
This recall involves products made in the Abbott Columbus facility but does not include any other liquid or powder formula brands or other nutrition products produced in the factory other than the ones named.
If you purchased one of the recalled products, do not use it, Abbott says.
“This recall equates to less than one day’s worth of the total number of ounces of infant formula fed in the U.S. and is not expected to impact the overall U.S. infant formula supply,” the Pedialyte and Similac recall states.
The products included in the recall were distributed primarily to hospitals and to some doctors’ offices, distributors and retailers in the United States, including Puerto Rico.
In addition, Abbott sent one lot of products to Barbados, Bermuda, Colombia, the Dominican Republic, Haiti, Jamaica, St. Croix and St. Thomas. Two lots were sent to Canada, Curacao, Panama and Trinidad and Tobago.
Have you been affected by the nationwide baby formula shortage? Let us know in the comments!
Don’t Miss Out!
Check out our list of Class Action Lawsuits and Class Action Settlements you may qualify to join!
Read About More Class Action Lawsuits & Class Action Settlements:
- FDA investigation into baby formula shortage concludes ‘shortcomings’ hampered agency response
- Ensure class action alleges nutrition drinks falsely advertised as healthy food choice
- Kroger class action alleges Simple Truth Organic Rice Rusks Baby Teething Wafers contain toxic metals
- Baby Trend’s Cityscape Travel Jogger Strollers recalled due to fall, injury hazards














2 thoughts onSimilac, Pedialyte recall announced due to potential spoilage
Please add me
Add me