When the Penumbra JET 7 Xtra Flex catheter was introduced to the market for people who suffered certain types of strokes, the manufacturer touted the device as its “most advanced technology.” However, the California-based medical device company is now alerting healthcare professionals that the JET 7 Xtra Flex catheters should be removed from health care facilities and pulled from the market.
On Dec. 15, 2020, after Penumbra catheter complications were attributed to 20 fatalities, the company issued an urgent voluntary recall acknowledging that the distal tip on the device can potentially be damaged — resulting in the risk of fatality or serious injuries to patients.
What Does the Penumbra Catheter Do?
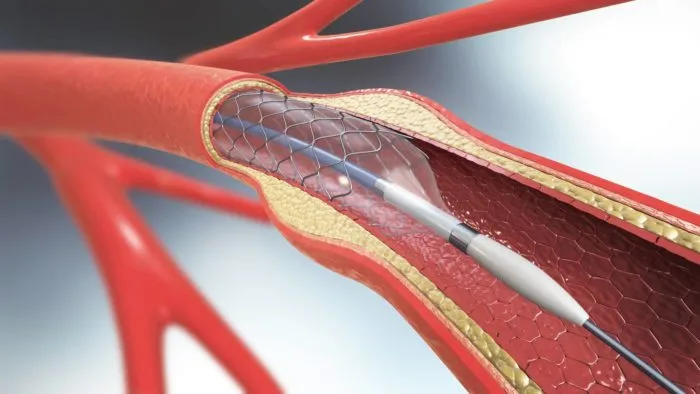
The Penumbra catheter JET 7 Xtra Flex was designed to be used in patients who have suffered the effects of acute ischemic stroke. Specifically, the device was meant to be used in a medical procedure called a suction thrombectomy to remove blood clots in veins and arteries. The catheter would be inserted in the patient at the groin or wrist to restore blood flow from the arteries to the brain.
Are Penumbra Catheters Commonly Used?
Penumbra catheters have long been used in hospitals. In fact, approximately 80% of suction thrombectomies nationwide have involved the use of one of Penumbra’s devices. Despite the potential safety issue, they are still expected to be used in 65% of suction thrombectomies this year. According to statistics, hospitals spent $168 million on the Jet 7 Xtra Flex in 2019, accounting for over 30% of the company’s sales that year.
What Are the Problems With the Penumbra Catheter?
Designed to treat individuals who have suffered strokes, the Penumbra catheter known as the JET 7 Xtra Flex raised a number of concerns over the last several months that it may have been defective. Since its June 2019 approval by the U.S. Food and Drug Administration (FDA), Penumbra distributed some 30,000 devices. Over the course of time the catheter spent on the market, it has been attributed to 18 deaths — 15 in the U.S. and three in Japan.
While the device was widely acclaimed when it was announced at an industry conference held in July 2019, some surgeons have since claimed that the catheter would sometimes fray or break during procedures. Medical device reports sent to the FDA also revealed other failures including ballooning, breaking, and complete separation. Exposure of internal support coils near the distal tip was also an issue reported in connection with the device.
At the FDA’s prompting, Penumbra alerted doctors in July 2020 with a “Notification to Healthcare Providers” that it received reports about the catheter’s distal tip expanding or rupturing “during injection of contrast media.” While the manufacturer didn’t deny the allegations of reported problems with the distal tips, it stated in the notification that the device had “not been tested for compatibility with other manufacturer’s revascularization devices.”
After issuing the warning, the FDA continued to receive reports from doctors in connection with Penumbra catheter complications and the device’s failures. Due to the evidence of risks associated with the device, the FDA requested the company issue a voluntary recall.
 What Are the Alleged Penumbra Catheter Complications?
What Are the Alleged Penumbra Catheter Complications?
According to the FDA’s Manufacturer and User Facility Device Experience database, known as MAUDE, there were 11 deaths linked to the Penumbra catheter JET 7 Xtra Flex between January 2020 and July 2020. All of these reports involved a problem with the catheter’s distal tip expanding or fracturing. Since July, seven more deaths were reported to have occurred. Most of the reports detailed at least one arterial rupture occurring, including many involving the artery that supplies blood flow to the eyes and brain.
In total, the FDA received more than 200 medical device reports concerning the device.
In addition to fatality, the FDA stated that doctors have reported serious injuries in connection with Penumbra catheter complications such as vessel damage, hemorrhage, and cerebral infarction — tissue damage in the brain due to loss of oxygen.
The Penumbra Catheter Recall
After determining the device could pose a risk of injury or fatality, Penumbra issued an urgent voluntary recall. In addition to the JET 7 Extra Flex catheter, the recall also applies to the JET 7MAX configuration, which was approved by the FDA in February 2020.
The specific product names Penumbra listed in its recall notification include:
- Penumbra JET 7 Reperfusion Catheter and Penumbra Hi-Flow Aspiration Tubing, Catalog Number 5MAXJET7KIT
- Penumbra JET 7 Reperfusion Catheter and Penumbra Hi-Flow Aspiration Tubing, Catalog Number 5MAXJET7L138KIT
- Penumbra JET 7 Reperfusion Catheter, Catalog Number 5MAXJET7
In its notice, Penumbra admitted the device’s potential defect, stating, “Distal tip damage in conjunction with pressurization or contrast injection may result in potential vessel damage, and subsequent patient injury or death.”
The FDA stated that those who had undergone successful treatments with the device are not affected by the recall.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Free Penumbra Catheter Defective Product Case Evaluation
If you qualify, an attorney will contact you to discuss the details of your potential case at no charge to you.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
E-mail any problems with this form to:
[email protected].
Oops! We could not locate your form.












