 Intrauterine devices continue to grow in popularity in the United States as the U.S. Food & Drug Administration (FDA) approves Skyla, the first new IUD to be released into the country in 13 years. Skyla is manufactured by Bayer Pharmaceuticals.
Intrauterine devices continue to grow in popularity in the United States as the U.S. Food & Drug Administration (FDA) approves Skyla, the first new IUD to be released into the country in 13 years. Skyla is manufactured by Bayer Pharmaceuticals.
While most medical experts find it no surprise that women continue to opt for this method of contraception, it does surprise some experts that the number has increased over the past few years despite the number of injury reports. Popular intrauterine devices that have already been released in America include the Mirena IUD and Paragard. Both come with the same risk of device migration and other complications upon implantation. It remains to be seen if Bayer’s Skyla will have the same number of complications.
IUDs have primarily grown in popularity because of their convenience and ease of use. Unlike oral contraceptives, IUDs do not require the patients to follow a strict schedule and only need to be replaced every few years. Additionally, some studies have suggested that these devices are actually more effective than birth control pills.
Despite the amount of success these devices have garnered, some physicians are reluctant to prescribe IUDs. In March 2012, a study published in Obstetrics & Gynecology found that the concern of IUD complications prevented many doctors from recommending these devices as contraceptives for women.
Additionally, in a survey conducted by the researchers from the U.S. Center for Disease Control and Prevention (CDC) found that 30 percent of all family doctors, OB/GYNs, nurses and other health care professionals thought that intrauterine devices were not safe for women who had not had children and called the general safety of the device into question. These concerns have led some doctors to only provide IUDs on an “occasional” basis, usually when their patient expressed preference for the option.
Overview of Intrauterine Device Complications
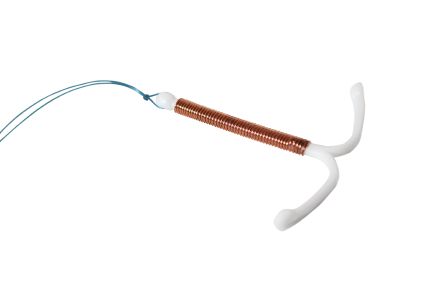
Currently, the most popular intrauterine device in the United States is the Mirena IUD, manufactured by Bayer Pharmaceuticals. The Mirena IUD is a small T-shaped device made of biologically safe plastic, which releases levonorgestrel after it is implanted in the uterus. Like all intrauterine birth control devices, it is a long-term birth control device, able to last up to five years. After it was approved by the FDA in 2000, Bayer allegedly aggressively marketed the device as a difficulty-free birth control option.
Most women opted for the Mirena IUD based on its ease of use and its nearly instant and long-lasting effectiveness. However, soon after its release, many patients started reporting complications of IUD perforation, migration and embedment. Reportedly, the device could migrate from the uterus and implant itself in another organ, causing a variety of problems.
Since its release in 2000, more than 70,000 Mirena IUD injuries have been reported to the FDA, including 5,000 cases of device migration, and 1,322 reports of a punctured uterus.
Help for Victims of Mirena IUD Complications
If you or a loved one had a Mirena IUD inserted after January 1, 2000 or later and had to have surgery – or will be required to have surgery – to have it remove because it migrated, you may be eligible to take legal action against the manufacturer. Filing a Mirena IUD lawsuit may help you recover compensation for medical bills, pain and suffering and other damages. See if you qualify by filling out the short form at the Mirena IUD Injury Class Action Lawsuit Settlement Investigation.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.















One thought on IUD Popularity Growing Despite Concerns of Mirena IUD Injuries