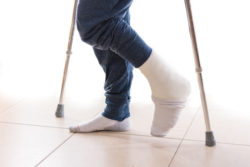
 A man filed an Invokana amputations lawsuit against the drug’s manufacturers after he allegedly suffered amputation of toes due to diabetes and other side effects he says he was never warned about.
A man filed an Invokana amputations lawsuit against the drug’s manufacturers after he allegedly suffered amputation of toes due to diabetes and other side effects he says he was never warned about.
Plaintiff James A. filed the Invokana amputations lawsuit in New Jersey federal court joining a growing multidistrict litigation (MDL) against the company. The lawsuit was filed on March 16, 2018.
According to the Invokana amputations lawsuit, James, a resident and citizen of the state of Iowa, says that he was prescribed Invokana and began taking the medication for diabetes treatment in July 2016.
He says that he began taking Invokana to reduce his blood sugar level and relied on the drug manufacturer’s claims that Invokana was “safe and effective for the treatment of diabetes.”
However, he alleges he suffered several adverse side effects, including Invokana amputations. James alleges he had to undergo amputation of toes due to diabetes, and that this amputation was the “direct and proximate result” of him being prescribed Invokana.
His amputation of toes due to diabetes occurred on Oct. 14, 2016, including the removal of the fourth digit of his right foot, and later he suffered amputation of toes due to diabetes on the third digit of his left foot. Further, alleges the plaintiff, on March 10, 2017, he suffered from additional amputation on the third digit of his right foot.
According to the Invokana amputations lawsuit, James’ injuries were preventable and resulted from the lack of proper safety studies. The complaint states that the drug maker’s suppression of information resulted in “serious and life-threatening risks” and they further engaged in the “willful and wanton failure to provide adequate instructions,” and the “willful misrepresentations concerning the nature and safety of Invokana.”
The Invokana amputations lawsuit was filed on multiple counts including strict liability, manufacturing defect, design defect, failure to warn, negligence, breach of express warranty, breach of implied warranty, negligent misrepresentation, fraudulent misrepresentation, unjust enrichment, fraud, Violation of Consumer Protection Laws, and punitive damages.
James demands a trial by jury.
Overview: Invokana Amputations
Invokana (canagliflozin) is a type-2 diabetes medication prescribed to lower patients’ blood sugar levels. It is prescribed in combination of diet and exercise.
On March 29, 2013, Janssen Pharmaceuticals announced Invokana’s approval by the U.S. Food and Drug Administration (FDA) for treatment of adults with type-2 diabetes. It also belongs to a class of drugs known as sodium-glucose contransporter-2 (SGLT2) inhibitors.
However, there have been several adverse effects associated with Invokana, including a severe complication linked to the drug that was announced by the FDA in May 2017.
The federal agency’s warning states “based on new data from two large clinical trials, the U.S. Food and Drug Administration (FDA) has concluded that the type-2 diabetes medicine canagliflozin (Invokana, Invokamet, Invokamet XR) causes an increased risk of leg and foot amputations.”
“We are requiring new warnings, including our most prominent Boxed Warning, to be added to the canagliflozin drug labels to describe this risk.”
The Invokana Amputations Lawsuit is Case No. 3:18-cv-03732-BRM-LHG, in the U.S. District Court for the District of New Jersey. The Invokana Lawsuit is in re: Invokana (Canagliflozin) Products Liability Litigation, MDL No. 2750 in the U.S. District Court for the District of New Jersey.
In general, Invokana and Invokamet lawsuits are filed individually by each plaintiff and are not class actions.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Invokana Class Action Lawsuit Investigation
If you or a loved one suffered ketoacidosis or lower extremity amputation after taking Invokana, Invokamet, or Invokamet XR, you may have a legal claim. See if you qualify to pursue compensation and join a free diabetes medication class action lawsuit investigation by submitting your information for a free case evaluation.
An attorney will contact you if you qualify to discuss the details of your potential case.
Please Note: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client, if you qualify, or getting you dropped as a client.
Oops! We could not locate your form.













3 thoughts onMan Files Invokana Amputations Lawsuit Over Toe Amputation