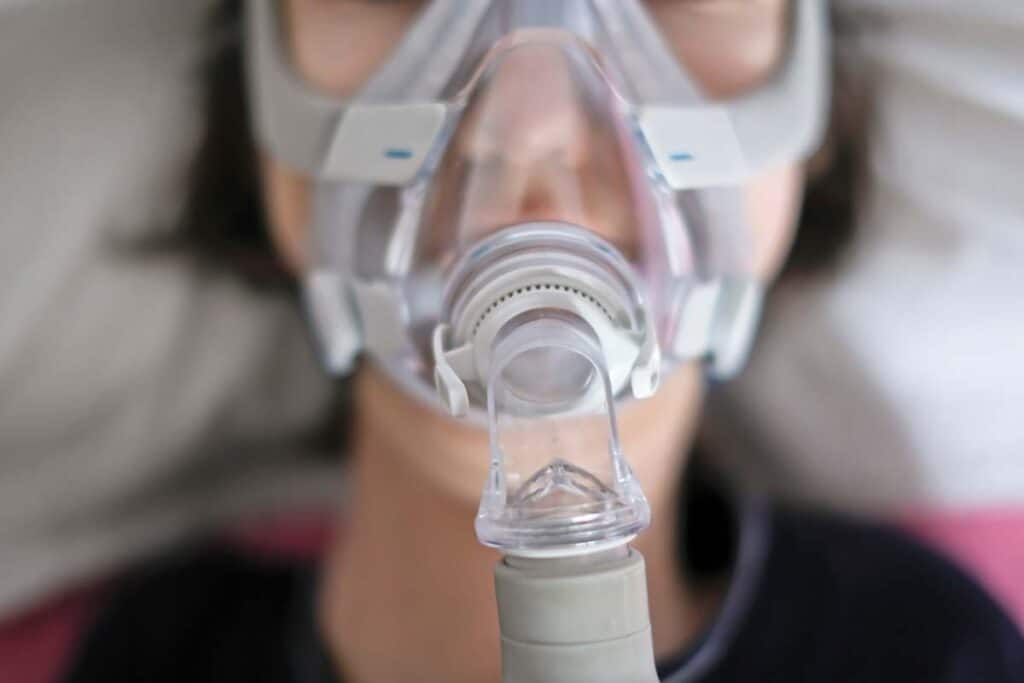
Philips CPAP overview:
- Who: Philips agreed to stop selling its CPAP and BiPAP machines in the United States while it works to comply with a consent decree with the Department of Justice and Food and Drug Administration.
- Why: Philips faces allegations of defective foam in its CPAP, BiPAP and other respiratory devices that exposed patients to dangerous chemicals.
- Where: The announcement affects U.S. Philips customers.
Philips agreed to stop selling its CPAP and BiPAP machines in the United States while it works to comply with a consent decree with the Department of Justice and Food and Drug Administration.
In December, the company agreed to a $494 million class action lawsuit settlement to resolve claims it included defective foam in its CPAP, BiPAP and other respiratory devices it recalled in June 2021. The foam allegedly exposed patients to dangerous chemicals.
Philips will continue to sell new CPAP and BiPAP devices outside of the United States but only service existing devices and supply accessories, consumables and replacement parts within the United States, according to the consent decree, the company says in its Fourth Quarter and Annual Results 2023.
“Until the relevant requirements of the consent decree are met, Philips Respironics will not sell new CPAP or BiPAP sleep therapy devices or other respiratory care devices in the U.S.,” Philips says in the document.
Philips spent €363M on remediation in fourth quarter of 2023
Philips says that more information about the consent decree will become available when it’s approved by the court.
The company divulged its multiyear plan included €363 million euro in the fourth quarter of 2023 for remediation, inventory write-downs and contract provisions.
“In 2024, Philips expects around 100 basis points of costs that relate to remediation activities and disgorgement payments for Philips Respironics sales in the U.S.,” the company says in the results report.
Philips shares were down 8.3% in the hours after the company announced the decision, Reuters writes, quoting ING analyst Marc Hesselink as calling it “very punitive.”
“We believe it will be very difficult for Philips to recover its U.S. Respironics market position,” Hesselink tells Reuters.
However, Philips Chief Operating Officer Roy Jakobs says he is confident about the company’s future.
“Based on our ongoing actions to enhance execution, we expect further performance improvement in 2024,” Jakobs says in the results report.
Do you have a Philips CPAP or Philips BiPAP device? Let us know in the comments.
Don’t Miss Out!
Check out our list of Class Action Lawsuits and Class Action Settlements you may qualify to join!
Read About More Class Action Lawsuits & Class Action Settlements:















15 thoughts onPhilips stops sale of CPAP, BiPAP machines amid FDA investigation
I have 2 machines that were recalled. How do I get into the lawsuit?
I use the Philips CPAP . I’m not feeling very confident in this machine. Going to call my Insurance company right now.
Since Phipis can not sell products in US until the FDA releases them, does that mean that all the replacement devices they sent out are still defective? And now I received a notice in the mail of a Data breach in their MOVEit transfer application they use.
I have been using a Philips CPAP for years