
On May 14 of this year, the Food and Drug Administration issued warning letters to two different breast implant manufacturers due to concerns over the companies’ failure to comply with specific regulatory requirements. One of the letters was regarding Allergan implants which, according to Fortune, have been linked to a type of cancer known as breast implant-associated anaplastic large cell lymphoma (BIA-ALCL). As of June 3, more than 33 women have died from BIA-ALCL.
In response to the FDA’s letters, Allergan issued a recall on their BIOCELL textured breast implants. Unfortunately, due to a lack of medical device tracking, many women don’t know that their Allergan implants have been recalled. The manufacturer has announced they will be launching a digital and social media campaign meant to alert patients about the recall.
Allergan Implants Recall

“This is a really unusual step, and it does show that the FDA understands that the breast implant safety advocates have established communication mechanisms that they haven’t,” former FDA manager and medical device failure expert Madris Tomes told Fortune.
So far, Allergan reports that there are at least 52,000 implants that they can’t locate. To help combat the issue, Allergan is encouraging patients to register their implants on the company’s site. The registration is available for U.S. patients only despite the fact that the recall is in effect worldwide. The registration form does include an option for patients who aren’t sure what kind of implant they may have.
Under the recall, all unused implants may not be used or sold, but those with Allergan implants who haven’t experienced any negative effects should consult with their doctor. Implant removal may not be needed.
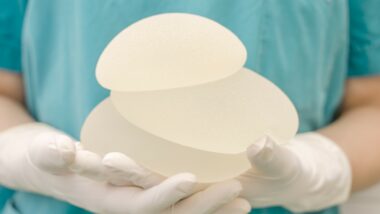
Allergan Implants Health Risks
According to Allergan, the BIOCELL implants were recalled due to “an association between breast implant-associated anaplastic large cell lymphoma (BIA-ALCL)” and Allergan implants of this type. BIA-ALCL is a serious condition that can be fatal, especially if left untreated. However, Allergan reports that less than 0.03 percent of BIOCELL patients have developed BIA-ALCL.
BIA-ALCL is a non-Hodgkin’s lymphoma, meaning it is a cancer of the immune system. This cancer is generally found in the scar tissue and fluid in the area around the Allergan implants, but there is a chance it can spread throughout the body. Symptoms of BIA-ALCL include breast enlargement, persistent pain, hardening, lumps in the breast or armpit, or fluid buildup surrounding the implant.
If you have Allergan implants and have experienced any of these symptoms, contact your medical provider as soon as possible. Leaving BIA-ALCL untreated can lead to death. Treatment generally involves removing the implant and any scar tissue.
Should You File an Allergan Implants Lawsuit
If you received Allergan implants only to later develop breast implant-related cancer, you may be eligible to file a lawsuit against Allergan. Currently, the company is offering up to $7,500 for out-of-pocket surgery costs for patients who developed BIA-ALCL as well as $1,000 towards diagnostic testing.
However, Fortune reports that these amounts aren’t enough to cover the cost of the procedures. The company is now facing 48 lawsuits, some of which are class action lawsuits that other patients may be able to join. Seeking legal action may be an effective way to access compensation for any medical expenses you may have incurred.
Join a Free Breast Implants Side Effects Lawsuit Investigation
You may qualify for this breast implant investigation under these circumstances:
- You were implanted with BIOCELLE or Natrelle textured breast implants;
- You were diagnosed with Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA ALCL); or
- You have had revision surgery after learning about this cancer risk.
Fill out the form on this page for a free case evaluation by a breast implants injury attorney.
This article is not legal advice. It is presented
for informational purposes only.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Breast Implants Side Effects Lawsuit Investigation
If you qualify, an attorney will contact you to discuss the details of your potential case at no charge to you.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
E-mail any problems with this form to:
[email protected].









One thought on Allergan Implants Recall Campaign Initiated