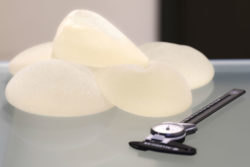
 Allergan has recalled its Biocell textured breast implants at the request of the U.S. Food and Drug Administration (FDA), citing the agency’s breast implant illness statistics from Medical Device Reports.
Allergan has recalled its Biocell textured breast implants at the request of the U.S. Food and Drug Administration (FDA), citing the agency’s breast implant illness statistics from Medical Device Reports.
The FDA has received reports of 573 incidents of a rare cancer known as breast implant-associated anaplastic large cell lymphoma (BIA-ALCL). Of those, 481 were linked to the Biocell implants. Thirty-three reported deaths are associated with the illness. The type of breast implant was determined in 13 of the deaths, and 12 of those victims who died had Allergan implants.
The textured breast implants are reportedly not as often used in the U.S. as they are in other countries, said the FDA. Only five percent of the breast implants sold in the U.S. are macro-textured like the Biocell brand.
Allergan said the voluntary worldwide recall affects its saline-filled and silicone-filled textured breast implants and tissue expanders. These products will no longer be distributed or sold, and healthcare providers are asked to return any unused Biocell textured implants and tissue expanders to Allergan.
Breast Implant Illness Statistics Prompt Recall
Dr. Jeffrey Shuren, director of the FDA’s Center for Devices and Radiological Health, told the New York Times, “The data regarding deaths was particularly informative of our decisions.”
BIA-ALCL is not a breast cancer, but is a type of non-Hodgkin’s lymphoma, a cancer of the immune system. Instead of attacking breast tissue, BIA-ALCL originates in the scar tissue capsule next to the implant. The scar tissue is the body’s natural response to a foreign object, but why anaplastic large-cell lymphoma might develop there is unknown.
The type of contents within the implant, silicone or saline, has not been an issue in whether a patient develops BIA-ALCL. The covering of the implant is either smooth or textured, but the type of texture varies. The idea behind making the implants textured was to encourage the scar tissue to better adhere to the surface, resulting in a more secure anchoring of the implant. It’s the very textured Biocell implants that have been most often linked with BIA-ALCL.
The FDA initially became suspicious of the potential link between breast implants and ALCL in January 2011. At that time, research identified 34 women diagnosed with the rare cancer that had developed around a breast implant.
The FDA said that the recalled Biocell macro-textured implants are six times more likely to cause ALCL than any other textured implant sold in the U.S., where hundreds of thousands of women have the implants, according to the Director of the FDA’s Office of Surgical and Infection Control Devices Dr. Binita Ashar. She presented those figures at a news conference regarding the Allergan recall on July 24.
Even though the risk of developing BIA-ALCL is low, the FDA wrote in a safety communication to patients that “at the present time, we believe all individuals who are considering a breast implant of any type be informed of the risk of developing” BIA-ALCL.
The New York Times spoke with Raylene Hollrah, an ALCL survivor who was diagnosed in 2013 and has been advocating for breast implant recalls. She spoke of knowing women who have developed BIA-ALCL who have different implants, not Allergan’s Biocell. She hears of women who have been incorrectly diagnosed or have had their concerns dismissed by their doctors.
Women who develop swelling and accumulated fluid round a breast implant should have the fluid drained. Diagnostic tests will be run to determine if ALCL is present.
Join a Free Breast Implants Side Effects Lawsuit Investigation
You may qualify for this breast implant investigation under the following circumstances:
- You were implanted with textured breast implants;
- You were diagnosed with Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA ALCL); and/or
- You’ve suffered from any illness you believe is related to the implants.
Fill out the form on this page for a free case evaluation by a breast implants injury attorney.
This article is not legal advice. It is presented
for informational purposes only.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Breast Implants Side Effects Lawsuit Investigation
If you qualify, an attorney will contact you to discuss the details of your potential case at no charge to you.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
E-mail any problems with this form to:
[email protected].






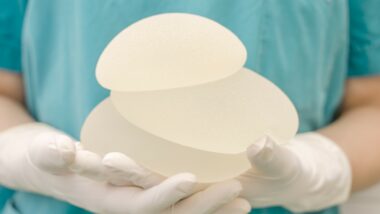



One thought on Breast Implant Illness Statistics Prompt Recall of Allergan Biocell Textured Implants