 More than two decades after the first power morcellation devices hit the market, the FDA approved the first surgical bag to contain tissues.
More than two decades after the first power morcellation devices hit the market, the FDA approved the first surgical bag to contain tissues.
Still, the federal agency cautions that the containment bags have not been proven to reduce the risk of disseminating power morcellator cancer and should only be used in a limited number of laparoscopic gynecological surgery cases.
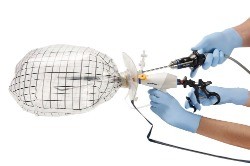
About the PneumoLiner Containment Bag
The PneumoLiner is a containment bag and a tube-like plunger designed to enclose an organ and inflate, which creates a working space that allows for visualization during power morcellator surgery and helps prevent breaking the bag with the tip of the morcellation device or other surgical tools.
The FDA says the PneumoLiner is intended as an alternative to more invasive surgery only “in a very limited patient population for whom power morcellation may be an appropriate therapeutic option — and only if patients have been appropriately informed of the risks.” The agency also placed a similar “Black Box” warning on the PneumoLiner like the one that appears on on power morcellators.
According to the FDA statement, the device was tested in laboratories to simulate worst-case scenarios.
“The containment bag was found to be impermeable to substances that were similar in molecular size to tissues, cells and body fluids, and other testing determined that the inflated bag provided adequate space for surgeons to perform morcellation with good visualization. Stress testing to evaluate the bag’s mechanical strength demonstrated that the device could withstand forces in excess of those expected to occur in actual clinical use.”
However, the FDA is requiring the manufacturer of the device to warn patients and their healthcare providers that it has not been proven to reduce the risk of spreading power morcellator cancer during these procedures.
Power Morcellation Risks
Power morcellation devices are surgical tools that are used during fibroid surgery and hysterectomies to grind up uterine tissue into pieces that can be removed through small incisions. In the past, hand-operated morcellation devices were almost always used with surgical bags.
When power morcellators hit the market in the 1990s, the bags were rarely used because they were cumbersome and most surgeons though the risk of spreading power morcellator cancer was low.
That all changed two years ago, when the FDA warned that 1 in 350 women undergoing a hysterectomy or fibroid surgery actually has undiagnosed uterine sarcoma, and 1 in 500 has highly-aggressive leiomyosarcoma.
The problem is that morcellators often leave behind tiny pieces of tissue. If the pieces contain cancerous cells, morcellators can spread aggressive power morcellator cancer throughout a woman’s abdomen and significantly increase her risk of death.
Despite the FDA’s approval of the containment bag, at least two U.S. health centers will not be using PneumoLiner bags. Both Rochester Regional Health and UR Medicine banned the use of power morcellation devices last year after three patients reportedly underwent laparascopic surgery where their undiagnosed cancer spread through the use of morcellation devices.
“This new device does not change our position on the risks associated with power morcellation,” Dr. William Maisel, chief scientist at the FDA’s Center for Devices and Radiological Health, said in the news release announcing the marketing of PneumoLiner. “We are continuing to warn against the use of power morcellators for the vast majority of women undergoing removal of the uterus or uterine fibroids.”
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The morcellation cancer attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. [In general, morcellator cancer lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Morcellation Cancer Class Action Lawsuit Investigation
If you or a loved one were diagnosed with cancer in the uterus, pelvis or abdomen within two years of undergoing surgery for a myomectomy (removal of fibroids), hysterectomy (removal of the uterus), oophorectomy (removal of the ovaries), or salpingectomy (removal of fallopian tubes), you may have a legal claim. See if you qualify by filling out the short form below.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Oops! We could not locate your form.












