Stryker Hip Implant Lawsuits Grow Following Recall
By Andrea Gressman
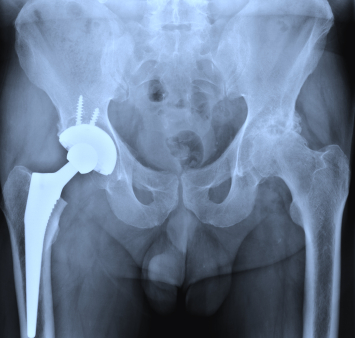 Following complications arising against two Stryker hip implants, there are a number of users that have chosen to file Stryker hip implant lawsuits. Because of these Stryker lawsuits, the manufacturer of these hip replacement products has issued a recall for its Rejuvenate hip implant systems. The complaints numbered in the hundreds, which compelled the FDA to investigate and issue a warning to the company adding another reason for the manufacturer to issue a recall.
Following complications arising against two Stryker hip implants, there are a number of users that have chosen to file Stryker hip implant lawsuits. Because of these Stryker lawsuits, the manufacturer of these hip replacement products has issued a recall for its Rejuvenate hip implant systems. The complaints numbered in the hundreds, which compelled the FDA to investigate and issue a warning to the company adding another reason for the manufacturer to issue a recall.
Stryker Hip Implant Side Effect Leads to Bone Fractures and Squeaking Joints
FDA had initially approved the Stryker Trident hip implant in 2003 but negative side effects started being reported by 2005. This ceramic-on-ceramic device received numerous failure complaints and replacement requests in 2005 causing a concern for the company. A few other complications were reported with its ceramic part positioning and improper seating that caused joints to squeak and made bones prone to fracturing. As these side effects became more prevalent, Stryker hip implant lawsuits were filed against the company as well.
Only after such allegations were brought to light in 2005 did the FDA finally inspect the Cork, Ireland manufacturing facility of Stryker. There they found plenty of violations in manufacturing practices along with the inability to establish repair procedures and adequate risk analysis.
In 2007, Stryker received a second warning regarding its New Jersey plant when it was found to contain clusters of the fatal Staphylococcus bacterial infection. This caused a complete recall and on January 22, 2008 Stryker did follow through with that recall of the Trident Acetabular Cup System and the Trident Hemispherical Cup System on January 2008.
Bone Loss and Metallosis Among Stryker Rejuvenate Hip Implant Side Effect Issues
The Stryker Rejuvenate Modular system is another device that the FDA approved for marketing in June 2008. This metal-on-metal hip replacement system was made without any plastic or ceramic parts. It was supposed to be years ahead of the competition with it being marketed as a flexible hip replacement option. However, it was met with a number of complaints following its launch. A number of the complaints claimed that the neck and stem components wore against each other and caused a number of metal hip implant side effects, including the following:
* Bone damage
* Bone loss
* Metallosis
* Premature hip implant failure
* Pseudotumors
* Tissue damage
First, a Canadian recall was issued and that led the FDA to issue another warning to the company and a recall of the Stryker Rejuvenate hip implant device was issued on July 6, 2012. A number of Stryker hip implant lawsuits have been filed since that time.
Stryker Hip Implant Lawsuits Continue to Rise
Many of those who have already suffered from negative side effects caused by a Stryker hip implant have filed Stryker hip replacement lawsuits seeking compensation their injuries caused by Stryker’s negligence. Both federal and local courts have had Stryker hip implant lawsuits filed in their courts. Considering the fact that Rejuvenate Modular system has only recently been recalled, the Stryker lawsuits filed against the company will continue to grow. If you have experienced negative side effects due to the Stryker hip implant consider filing a Stryker hip implant lawsuit of your own. There are a number of options for you to consider, including joint litigation, mass tort, or MDL.
There are a number of complaints regarding the Trident hip implant devices despite its recall. Subsequently, a number of Stryker hip implant lawsuits have been filed and consolidated in a number of states. One Stryker lawsuit consolidation in Massachusetts is a class action Stryker lawsuit consisting of around 40 plaintiffs and another in New Jersey has over 180 complainants in a centralized litigation.
If you have experienced one of more of the painful side effects caused by a Stryker hip implant, there is help for you. Visit the Stryker Metal Hip Implant Recall Class Action Lawsuit Investigation page and receive a free consultation with a qualified Stryker metal hip implant failure lawsuit attorney.
Updated November 15th, 2012
All medical device, dangerous drug and medical class action and lawsuit news updates are listed in the Drug and Medical Device section of Top Class Actions.
Top Class Actions Legal Statement









One thought on Stryker Hip Implant Lawsuits Grow Following Recall
Have Stryker Rejuvenate modular hip which has failed. I have very high cobalt/ chromium levels. Hip is being replaced.