 Two California women filed a GranuFlo lawsuit in March claiming that the kidney dialysis additive GranuFlo was the cause of their mother’s death.
Two California women filed a GranuFlo lawsuit in March claiming that the kidney dialysis additive GranuFlo was the cause of their mother’s death.
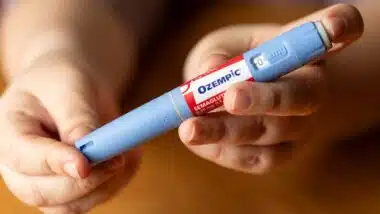
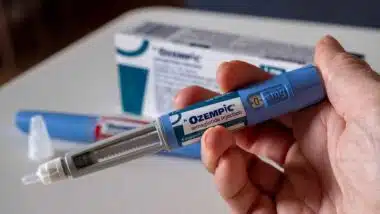
Deborah Lower and Constance Morgan filed the GranuFlo lawsuit in the U.S. District Court in Oakland, California on March 28. They are charging drug maker Fresenius Medical Care with wrongful death after their mother, Beaulahfay Riddie, who died while undergoing kidney dialysis and was treated with the drug. Her death was caused by metabolic alkalosis and cardiac arrest, which the lawsuit claims was directly caused by GranuFlo.
GranuFlo is used in kidney dialysis to help neutralize the acid in the blood by providing bicarbonate. For kidney dialysis treatment to work and do the job of filtering the blood that is normally done by the kidneys, an acid concentrate such as GranuFlo or NaturaLyte must be used. When the acid concentrate enters the bloodstream it is naturally converted to bicarbonate. Bicarbonate is how more than 90 percent of carbon dioxide takes form in the bloodstream. This keeps the blood from becoming too acidic — a job normally done by healthy kidneys.
The problem with GranuFlo and NaturaLyte is that they were not properly dosed an were linked to a potential rapid increase of bicarbonate, leading to a condition known as alkalosis — meaning that the blood has become too alkaline. If a person develops alkalosis, the risk of catastrophic heart side effects becomes 6 to 8 times more likely and can lead to sudden death.
This is what happened to Riddie. Prior to going into cardiac arrest, she had developed alkalosis and was experiencing severe pain and suffering.
The California sisters stated in their GranuFlo lawsuit that their mother’s death was the result of negligence and/or fraud. They claim that Fresenius knew about the dangers these additives caused and they failed to properly notify the doctors and clinics where the products were used of the potential dangers.
This claim is based on a memo Fresenius sent out in November 2011 to its own clinics — not all of the clinics that use GranuFlo and NaturaLyte — stating that GranuFlo was responsible for 941 deaths due to cardiac arrest at clinics in 2010. They notified all of the clinics where the acid concentrates were used in March 2012, but only after being questioned by the U.S. Food & Drug Administration upon receiving an anonymous copy of the memo.
In June 2012, the FDA issued a Class I recall of NaturaLyte and GranuFlo — the most serious recall they can issue that is restricted only for products that may result in severe injury or death.
Riddie’s daughters are asking for compensatory and punitive damages, pre- and post-judgment interest, costs and attorney fees in their GranuFlo lawsuit.
Several GranuFlo lawsuits were consolidated into multi-district litigation in the U.S. District Court, District of Massachusetts in April.
If you or someone you know experienced a negative side effect during kidney dialysis that you believe was caused by GranuFlo or NaturaLyte, legal options are available. Learn more and get a free consultation regarding a claim’s eligibility at the Kidney Dialysis Heart Attack, GranuFlo & NaturaLyte Recall Class Action Lawsuit Settlement Investigation. Experienced legal professionals acid concentrates played a role in your complications, so act now.
Updated May 2nd, 2013
All medical device, dangerous drug and medical class action and lawsuit news updates are listed in the Drug and Medical Device section of Top Class Actions.
Top Class Actions Legal Statement