A woman fearing she’s at greater risk of developing breast implant associated lymphoma has filed suit against Allergan.
A woman fearing she’s at greater risk of developing breast implant associated lymphoma has filed suit against Allergan.
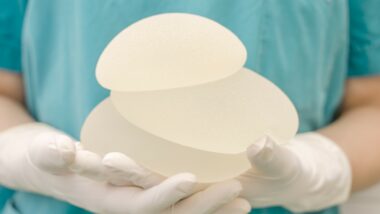
Plaintiff Lana Tauben said she received a set of Allergan implants in November 2016 that had to be removed in July of 2017 because she had developed persistent pain and swelling around them. The replacement set of implants she received were Allergan’s Natrelle 410 Highly Cohesive Anatomically Shaped Silicone-Filled Breast Implants FX.
The Natrelle implants were included in a recall by the U.S. Food and Drug Administration (FDA) on July 24, 2019, which issued a warning about highly textured breast implants, saying they could be linked to the development of breast implant associated anaplastic large cell lymphoma (BIA-ALCL), a rare blood cancer.
The FDA explains BIA-ALCL is not breast cancer, but is a non-Hodgkin’s lymphoma which affects the infection-fighting cells of the immune system. The cancer begins in the capsule of scar tissue that forms around the breast implant, separating the implant from the breast tissue.
The FDA has reported 573 cases of BIA-ALCL around the world, including 33 patient deaths. Of the 573 BIA-ALCL diagnoses, a total of 481 patients reportedly had Allergan breast implants at the time they were diagnosed.
Was Breast Implant Associated Lymphoma Hidden?
Tauben alleges in her Allergan lawsuit that “Allergan has known about the connection between its textured implants and the increased risk of developing BIA-ALCL since at least 2011.”
She further alleges that Allergan hid the evidence of problems, including implant ruptures and injuries, by including information of such incidents in Alternative Summary Reports (ASR), which are not mandated to be included in the FDA’s Manufacturer and User Facility Device Experience (MAUDE) database.
When the reporting loophole was discovered, the FDA changed its reporting requirements, which resulted in the number of reported breast implant-associated injuries increasing from 200 annually to 4,567 in 2017 and 8,242 in the first half of 2018. The FDA admitted the improvements in reporting these adverse events was the reason for the increase in numbers and not because the numbers reflected “a new public health issue.”
The FDA has required severe or unexpected events or injuries to be included in the MAUDE report system, but the FDA again came close to admitting breast implant manufacturers made the most of a loophole in the FDA’s reporting system. On May 2, 2019, the FDA said it was “announcing today a change in how breast implant manufacturers will file medical device reports with the FDA.” Potential symptoms of BIA-ALCL are not listed or reported.
The announcement went on to say the FDA was ending the ASR reporting program for all medical devices.
Because of her alleged increased risk of developing breast implant associated lymphoma, Lana believes that Allergan also should cover the costs of removing and replacing her breast implants.
Currently, Allergan is only offering free non-textured replacement implants and is not offering to cover the surgical costs or other related medical expenses. In the event a woman is diagnosed with BIA-ALCL, Allergan says it will pay a maximum of $1,000 for diagnostic procedures and a limit of $7,500 total for surgery and any necessary cancer treatments.
The Breast Implant Associated Lymphoma Lawsuit is Tauben. v. Allergan USA, Inc., et al., Case No. 2:19-cv-02257 in the U.S. District Court for the Central District of Illinois.
Join a Free Breast Implants Side Effects Lawsuit Investigation
You may qualify for this breast implant investigation under the following circumstances:
- You were implanted with textured breast implants;
- You were diagnosed with Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA ALCL); and/or
- You’ve suffered from any illness you believe is related to the implants.
Fill out the form on this page for a free case evaluation by a breast implants injury attorney.
This article is not legal advice. It is presented
for informational purposes only.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Breast Implants Side Effects Lawsuit Investigation
If you qualify, an attorney will contact you to discuss the details of your potential case at no charge to you.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
E-mail any problems with this form to:
[email protected].