
The Food and Drug Administration aims to better understand a possible connection between anaplastic large cell lymphoma and breast implants, particularly textured ones. This endeavor has led the FDA to qualify a self-administered questionnaire that will help patients dig into any symptoms that they might be experiencing in connection to their breast implants.
Recently, research has suggested that breast implants, specifically textured breast implants might be linked to BIA-ALCL, or breast implant-related anaplastic large cell lymphoma. However, this condition is not the only condition at issue. Patients sometimes refer colloquially to “breast implant illness,” which can describe a range of distressing symptoms including brain fog, fatigue, anxiety, depression, hair loss, autoimmune diseases, joint pain, inflammation, rash, and weight problems.
As researchers aim to better understand if or how these conditions could be connected to breast implants, they can now use a new questionnaire, the BREAST-Q Reconstruction Module, to learn more about patient experiences. The questionnaire aims to measure physical well-being, psychological well-being, sexual well-being, and a patient’s general satisfaction after having a breast implant surgery.
The FDA explains the benefits of this new analytical tool, noting that it can be used to evaluate breast implants, and can help the government make decisions around the regulation, sale, and approvals of breast implants. The FDA goes on to note that the BREAST-Q Reconstruction Module’s questionnaires are “scientifically validated,” indicating that they can provide valuable and useful data to the scientific community.
However, the use of these questionnaires are not required. Instead, an organization that sponsors a medical device, in this case a kind of breast implant, can choose whether or not to use these questionnaires in their product development.
Premier Research explains the role of a product sponsor, explaining that in medical device trials, a product sponsor helps organize and implement clinical trials. In the case of breast implants, a sponsor might examine data related to potential breast implant illness, would document the study, and make sure the study adhered to necessary regulation.

The FDA has also offered an update on existing data of statistics related to breast implant lymphoma. Now, the FDA has a recorded 733 BIA-ALCL cases around the world, and 36 deaths from the dais around the world. These numbers reflect cases reported through January 5, 2020. According to the FDA’s update, this is a 160 case and 3 death increase since the update issued in early July — a six month period. New cases could have developed since January.
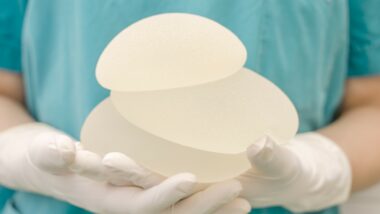
Unfortunately, research indicates that women with textured breast implants might be more at risk for breast implant lymphoma than those with smooth implants. Texas Medical Center explains that women sometimes prefer textured breast implants over smooth ones because they tend to stay in place better and look more natural, and come with less of a risk of scarring.
Of the 733 cases of BIA-ALCL reported to the FDA, 496 of those were textured. 209 cases reportedly did not specify what kind of surface the devices had.
Also concerning is that almost all of the cases of the disease were reportedly connected to implants made by one maker. The FDA states that of the reported 733 cases, 620 of these were allegedly Allergan implants. In the case of 47 breast implant lymphoma cases, the manufacturer was unknown.
As mentioned above, 36 patient deaths were reported in connection with BIA-ALCL. The FDA goes on to explain that of these dates, the manufacturer of the implants is only known in 16 cases. Of the 16 cases for which the manufacturer is known. 15 cases had Allergen implants when they were diagnosed with BIA-ALCL reports the FDA.
Join a Free Breast Implants Side Effects Lawsuit Investigation
You may qualify for this breast implant investigation under these circumstances:
- You were implanted with BIOCELLE or Natrelle textured breast implants;
- You were diagnosed with Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA ALCL); or
- You have had revision surgery after learning about this cancer risk.
Fill out the form on this page for a free case evaluation by a breast implants injury attorney.
This article is not legal advice. It is presented
for informational purposes only.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Breast Implants Side Effects Lawsuit Investigation
If you qualify, an attorney will contact you to discuss the details of your potential case at no charge to you.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
E-mail any problems with this form to:
[email protected].









One thought on FDA Approves New Patient Quiz, Updates Breast Implant Lymphoma Statistics