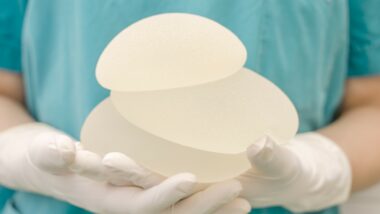
A Louisiana woman who survived breast cancer only to be diagnosed with a rare form of lymphoma linked to breast implants following a mastectomy is suing the implant manufacturer for failing to disclose problems to federal regulators.
In 2019, Christine D. was diagnosed with breast implant-associated anaplastic large cell lymphoma. or BIA-ALCL. In her Aug. 3 lawsuit, filed in U.S. District Court for the Western District of Louisiana, she says the implant manufacturer did not comply with U.S. Food and Drug Administration (FDA) requirements by failing to report “adverse events” that occurred with its implants, reporting Christine claims may have prevented her from ever getting the implants and developing cancer.
Allegations
According to Christine’s lawsuit, breast implant-maker McGhan, which was later purchased by another company before being sold to Allergan, the FDA in May 2000 granted pre-market approval for the implants Christine received.
As part of that approval, McGhan was required to conduct follow up studies of patients with the implants to further assess the long-term safety – to watch for possible links between the breast implants and cancer, among other things – and report the findings to the FDA.
Christine’s lawsuit says the company did not report “adverse events” from the post-market approval studies, which would have indicated the implants posed a risk of contributing to serious problems.
“(The company) knew of the manufacturing failures, and multiple risks associated with (the) implants design, and consciously responded by terminating the studies required … in favor of self-serving research that it could control,” the lawsuit claims. And “misrepresented the risks to the users, physicians, and regulatory agencies.”
Background
In 2000, Christine was diagnosed with bilateral breast cancer. She underwent a mastectomy and breast reconstruction, the civil complaint says. She and her doctor chose McGhan’s saline implants for the reconstruction.

Seven years later, Christine went to the doctor after noticing one of her breasts was enlarged. In October 2019, testing revealed she had breast implant-associated anaplastic large cell lymphoma.
Breast Implant-Associated Anaplastic Large Cell Lymphoma
Breast implant-associated anaplastic large cell lymphoma, or BIA-ALCL, is a cancer of the immune system, not breast cancer. It is very rare. Although an estimated 10 to 11 million women worldwide have gotten breast implants, fewer than 10 patients a year are diagnosed with the disease, according to the FDA.
Most cases of BIA-ALCL occur in those with textured implants, which are thought to cause more inflammation of the surrounding tissue than smooth implants, Cleveland Clinic says. Whether they are filled with saline or silicone does not seem to make a difference, studies have shown.
Allergies, reactions to implant materials, reactions to bacteria that grow on the surface of the implant, and genetics are contributing factors to breast implant-associated anaplastic large cell lymphoma, the hospital says.
The standard treatment is surgical removal of the breast implants and surrounding scar tissue.
Breast Implants and Cancer
In her lawsuit against Allergan, Christine accuses the manufacturer of misleading regulators and the public about the possible links between breast implants and cancer by omitting important information from its reports to the FDA.
Had she been aware of the risks, the plaintiff says she would not have chosen to get the implants and thus would have avoided getting BIA-ALCL.
The Allergan Breast Implants and Cancer Lawsuit is Christine Downey, et al., v. Allergan Inc., et al., Case No. 5:20-cv-00985, in the U.S. District Court for the Western District of Louisiana, Shreveport Division.
Join a Free Breast Implants Side Effects Lawsuit Investigation
You may qualify for this breast implant investigation under these circumstances:
- You were implanted with BIOCELLE or Natrelle textured breast implants;
- You were diagnosed with Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA ALCL); or
- You have had revision surgery after learning about this cancer risk.
Fill out the form on this page for a free case evaluation by a breast implants injury attorney.
This article is not legal advice. It is presented
for informational purposes only.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Breast Implants Side Effects Lawsuit Investigation
If you qualify, an attorney will contact you to discuss the details of your potential case at no charge to you.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
E-mail any problems with this form to:
[email protected].