 This month, plaintiff Sandra H. filed a complaint in a New Jersey federal court against Daiichi Sankyo Inc. and related companies named in a multidistrict litigation (MDL) case over Benicar’s colonic side effects.
This month, plaintiff Sandra H. filed a complaint in a New Jersey federal court against Daiichi Sankyo Inc. and related companies named in a multidistrict litigation (MDL) case over Benicar’s colonic side effects.
Defendants Daiichi Sankyo Inc. and Forest Laboratories LLC and their collective affiliates are the makers and promoters of Benicar, Benicar HTZ, Azor, and Tribenzor, pharmaceutical drugs for the treatment of hypertension or high blood pressure.
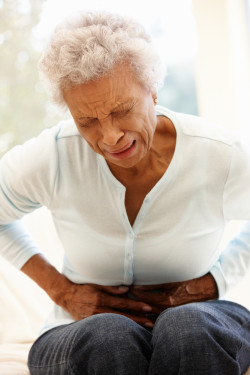
Sandra, a 65-year old citizen of the state of Florida, says she began taking Benicar for her hypertension around February 2008. Eventually, she began experiencing symptoms of severe colon distress.
Benicar colonic side effects stemming from its active ingredient, olmetsartan medoxamil, have been reporte in several individual cases. These side effects include chronic diarrhea, vomiting, nausea, colitis, enteropathy, malnutrition, weight loss and even kidney failure.
Sandra is requesting a trial by jury in her case and claims injury to herself because of high blood pressure treatment and Benicar colonic side effects.
She is raising 14 counts as presented in the MDL against the manufacturers and promoters of this drug including the following to name just a few: products liability for failure to warn and design defect, negligent misrepresentation, fraudulent concealment, and violation of the consumer protection act of the State of Florida.
The Food and Drug Administration’s Approval of the Benicar Family
The United States Food and Drug Administration (FDA) approved Benicar in the spring of 2002. Shortly following this approval came the go ahead for Benicar HTZ in the summer of 2003. In the fall of 2007, Azor tablets were sanctioned by the government.
Finally, in the summer of 2010, Tribenzor was similarly endorsed. This group of medications, often referred to as the Benicar family, all contain olmetsartan medoxamil and are identified as Angiotensin Receptor Blockers (ARBs).
After the FDA’s initial Benicar approval, the makers of the blood pressure drug put together an aggressive marketing campaign that attempted to sell it as a superiorly effective and safer treatment than other pharmaceutical ARB choices that were available.
In 2006, the FDA required Daiichi Sankyo and Forest Laboratories to backpedal on these claims, which the agency said were unproven and lacking evidence. The FDA also stated that printed promotional materials minimized and failed to acknowledge certain risks that are associated with the drug’s use.
Ultimately, the governing body required that the companies discontinue use of several promotional pieces and distribute corrective information to physicians and the public.
Benicar Colonic Side Effects
In the summer of 2013, the FDA issued a strong warning that the Benicar family containing olmetsartan medoxamil can cause intestinal problems that were commonly known as sprue-like enteropathy.
Weight loss accompanied by severely chronic diarrhea are among the common symptoms of this disorder. Sprue-like enteropathy in several cases took several months to years of treatment on Benicar to develop, but removal of the drug caused almost immediate remediation of symptoms, according to report submitted to the FDA.
The FDA insisted on a label change that included this warning. Sandra and plaintiffs like her believe that the company willfully concealed reports of Benicar colonic side effects and promoted the drug minimizing its risks while being fully aware of their existence.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or Benicar class action lawsuit is best for you. [In general, Benicar lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Benicar Class Action Lawsuit Investigation
If you or a loved one used Benicar for at least four months and were hospitalized for severe gastrointestinal problems, you may have a legal claim. Submit your information now for a free case evaluation.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Please Note: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client, if you qualify, or getting you dropped as a client.
Oops! We could not locate your form.












