In an attempt to force greater transparency in the medical device industry, the FDA is ending alternative summary reporting. This will end two decades of a practice that, for too long, has enabled the medical device and pharmaceutical industries to hide injury reports due to defective health care products – including surgical instruments and breast implants.
In an attempt to force greater transparency in the medical device industry, the FDA is ending alternative summary reporting. This will end two decades of a practice that, for too long, has enabled the medical device and pharmaceutical industries to hide injury reports due to defective health care products – including surgical instruments and breast implants.
Twenty Years of Secrets
The FDA’s alternative summary reporting, or ASR program was quietly instituted in October 1999. It allowed medical device manufacturers to submit reports of device malfunctions in a non-public FDA database, preventing the public and even practitioners from accessing them.
The ASR program got very little in the way of public notification. According to the exposé that appeared in Kaiser Health News in March 2019, many doctors and medical safety engineers knew nothing about it, and a former FDA commissioner said he was never aware that the ASR program even existed.
One surgeon interviewed for the story who experienced a serious malfunction with a surgical stapler said, “It seemed like a cover-up.”
Although deaths due to a defective device must still be reported through the Manufacturer and User Facility Device Experience (MAUDE) database, medical device manufacturers whose products had malfunctioned and caused injuries could still use the Alternative Summary Reporting program by making an “Exemption Request.”
Excuses
One FDA official told KHN that the ASR program was for issues “well-known and well-documented” within the agency, and that reforms had been put in place in 2017. That reform was a “voluntary summary reporting program” that covered as many as 5,600 devices.
Yet despite “reforms” and updates over the past two decades, the program has remained obscure and unavailable to the general public. It took investigative reporters several months before the FDA even acknowledged that Alternative Summary Reporting even existed.
An FDA spokesperson says that the exemptions allowed the agency to “… more efficiently review adverse events … and take action when warranted without sacrificing the quality of our review or the information we receive.” It is difficult to understand how this would be the case when it essentially has amounted to the FDA surrendering its oversight authority over the industry.
Critics say this obscurity and secrecy has resulted in hundreds of thousands of needless injuries, and has spurred massive amounts of litigation.
Alternative Summary Reporting Hid Millions of Incidents
In 2016 and 2017 alone, over 1.5 million injuries and malfunctions were reported through the ASR program. Another million or so reports filed over a 15-year period are still inaccessible by the general public.
It is possible to submit a Freedom of Information Act request about a given incident, but it can take as long as two years to get a response.
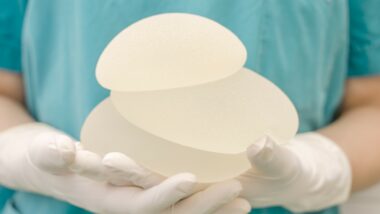
Breast Implant – Cancer Link Reports Hidden
Following publication of the KHN exposé, the FDA convened a panel for the review of injuries and diseases attributed to breast implants.
During that meeting, the committee addressed many reports of women who developed Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL) as a result of having textured breast implants. Incredibly, the FDA announced that it would not require the manufacturers of textured breast implants to issue a recall for the product; they currently remain on the market.
Join a Free Breast Implants Side Effects Lawsuit Investigation
You may qualify for this breast implant investigation under the following circumstances:
- You were implanted with textured breast implants;
- You were diagnosed with Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA ALCL); and/or
- You’ve suffered from any illness you believe is related to the implants.
Fill out the form on this page for a free case evaluation by a breast implants injury attorney.
This article is not legal advice. It is presented
for informational purposes only.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Breast Implants Side Effects Lawsuit Investigation
If you qualify, an attorney will contact you to discuss the details of your potential case at no charge to you.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
E-mail any problems with this form to:
[email protected].