Married Couple Sues AMS for SPARC Sling Vaginal Mesh Injuries
By Amanda Antell
 Virginia couple Tara Gray and Anthony Gray are suing American Medical Systems for injuries the female plaintiff sustained from their SPARC Sling System, a transvaginal mesh product designed to treat stress incontinence.
Virginia couple Tara Gray and Anthony Gray are suing American Medical Systems for injuries the female plaintiff sustained from their SPARC Sling System, a transvaginal mesh product designed to treat stress incontinence.
The plaintiff had the vaginal mesh product implanted on November 10, 2011, for personal health issues she was trying to correct. Soon after the transvaginal mesh was implanted she experienced severe mental and physical pain, suffered pain during intercourse, and found simple movements difficult.
Medical examination revealed that Gray was suffering from common side effects that many women experience after using vaginal mesh. The vaginal mesh complications were so severe that the plaintiff had to undergo revision surgery to fix the problem and still suffers from painful side effects.
While Gray’s situation is more than uncomfortable, her predicament with a transvaginal mesh implant is hardly uncommon. Thousands of women across America are suing American Medical Systems, Bard, Ethicon and other vaginal mesh manufacturers for negligence, concealing information, and failing to disclose vital safety information to patients or their physicians.
Gray states that had she known about the possible vaginal mesh complications she would endure, she would not have accepted the mesh into her body.
General Overview of Vaginal Mesh Complications
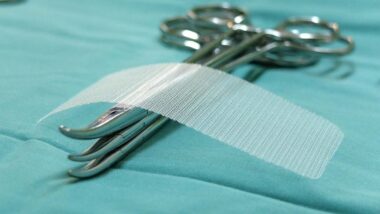
Vaginal mesh was originally created and designed to give women who suffered from pelvic organ prolapse (POP) and stress incontinence a permanent solution to their conditions.
Currently, the FDA categorizes transvaginal mesh products into four different categories, designated by their different uses:
- Non-Absorbable Synthetic: More than half of all mesh products approved by the FDA are in this group, with the main component of this group being plastic or polyester. About 91% of these mesh products are made from a popular, non-absorbable plastic called, polypropylene. These vaginal mesh products are designed for long-term or permanent treatments.
- Absorbable Synthetic: Unlike its cousin, this variety of transvaginal mesh loses strength and durability and is not meant to act as a permanent repair. Ideally, the patient’s growing tissue would grow over the mesh and would be strong enough to sustain device removal.
- Biologic: These transvaginal mesh products are made from animal tissue and are specifically meant to be implanted permanently into the human body. They are normally made from pig or bovine tissue.
- Composite: This vaginal mesh is made from a combination of any of the other categories.
While many women appreciate the ability to fix their problems in such a discrete way, it was soon discovered that these vaginal mesh implants could cause serious severe health problems, including mesh erosion and organ perforation.
Vaginal Mesh Lawsuits Grow
In 2011, there were 4,000 vaginal mesh injury reports submitted to the FDA, which was higher than the annual rate for the previous six years. Additionally, the FDA warned patients and surgeons against using transvaginal mesh for POP and stress incontinence treatment.
Nearly 23,000 lawsuits filed by women injured by vaginal mesh implants are pending in the U.S. District Court for the Southern District of West Virginia in a massive MDL.
The Gray’s vaginal mesh lawsuit is Tara Gray and Anthony A. Gray vs. American Medical Systems Inc., Case No. 13-cv-21606.
File a Vaginal Mesh Lawsuit Today
If you believe that you or a loved one has been the victim of a vaginal mesh injury, you have legal options. Please visit the Vaginal Mesh Sling Class Action Lawsuit Investigation. There, you can submit your claim for a free legal review and if it qualifies for legal action, a seasoned vaginal mesh lawyer will contact you for a free, no-obligation consultation. You will be guided through the litigation process at no out-of-pocket expenses or hidden fees. The vaginal mesh injury lawyers working this investigation do not get paid until you do.
All medical device, dangerous drug and medical class action and lawsuit news updates are listed in the Drug and Medical Device section of Top Class Actions.
Top Class Actions Legal Statement