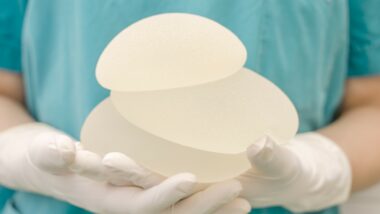
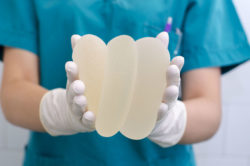
 The FDA recently required manufacturer Allergan to implement a textured breast implants recall based on reports of BIA ALCL, a rare type of cancer.
The FDA recently required manufacturer Allergan to implement a textured breast implants recall based on reports of BIA ALCL, a rare type of cancer.
“The U.S. Food and Drug Administration today took significant action to protect women from breast implant-associated anaplastic large cell lymphoma (BIA-ALCL) by requesting that Allergan, the manufacturer of a specific type of textured implant, recall specific models of its textured breast implants from the U.S. market due to the risk of BIA-ALCL,” the agency said in their July news release.
By the time the FDA made their request for the textured breast implants recall, other recalls had been made around the world in countries such as France, Canada, and Australia.
An Allergan spokesman responded to SELF in an email statement saying that they had initiated the FDA’s requested voluntary recall “as a precaution following notification of recently updated global safety information concerning the uncommon incidence of breast implant-associated anaplastic large cell lymphoma (BIA-ALCL) provided by the [FDA].”
Affected Products
The Allergan textured breast implants recall affects their BIOCELL textured breast implants and tissue expanders. Tissue expanders are used before breast augmentation or post-mastectomy reconstruction to stretch a person’s skin before implants are inserted.
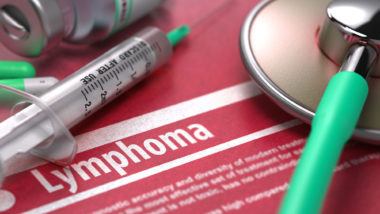
BIA ALCL is a rare form of cancer than can form around breast implants. After breast implants are implanted into the body, a scar tissue capsule forms around the implants during the healing process. BIA ALCL is a unique type of cancer because it forms only in this capsule. Luckily, this makes the treatment of the disease very simple as long as the cancer has not spread to other parts of the body.
Symptoms of BIA ALCL may include breast enlargement, pain, breast asymmetry, lumps, skin rash, breast hardening, fluid collection, and more.
BIA ALCL has been recorded with both silicone and saline implants and in patients with both smooth and textured implants. There is data to suggest that textured implants may pose an increased risk of BIA ALCL but smooth implants are also implicated.
ALCL was first identified by the FDA in 2011. At the time, the FDA was unable to determine risk factors for the rare cancer and called for more research. In 2016, the United Nations World Health Organization identified BIA ALCL as a type of T-cell lymphoma that can develop around the breast implant in the scar tissue capsule.
In February 2019, Health Canada announced that it would be updating its safety review of breast implants. In April 2019, the French National Agency of Medicine and Health Products Safety (ANSM) announced that it would be recalling “macro textured” breast implants from the market. In July 2019, the Australian Therapeutic Goods Administration (TGA) announced that it had proposed regulatory changes based on their assessment of textured breast implants.
The FDA has determined that they will not be completely recalling all textured breast implant like other foreign regulators due to the lack of evidence. They will reportedly continue to evaluate the risk profile of these devices and make regulatory changes based on any new evidence.
Join a Free Breast Implants Side Effects Lawsuit Investigation
You may qualify for this breast implant investigation under the following circumstances:
- You were implanted with textured breast implants;
- You were diagnosed with Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA ALCL); and/or
- You’ve suffered from any illness you believe is related to the implants.
Fill out the form on this page for a free case evaluation by a breast implants injury attorney.
This article is not legal advice. It is presented
for informational purposes only.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Breast Implants Side Effects Lawsuit Investigation
If you qualify, an attorney will contact you to discuss the details of your potential case at no charge to you.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
E-mail any problems with this form to:
[email protected].