 Another drug has joined the ranks of those that could cause Stevens Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN).
Another drug has joined the ranks of those that could cause Stevens Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN).
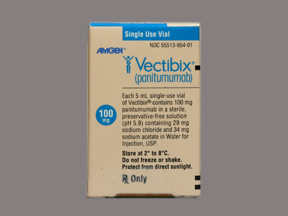
Amgen Canada, in consultation with Health Canada, in May released a safety information update regarding the use of Vectibix (panitumumab) and the risks of SJS/TEN.
Doctors and patients are advised to be observant, as the most common side effect of Vectibix is an “acne-like rash that mainly affects the head, chest, and back.” This is not the necessarily the same as an SJS reaction, but as many doctors have never seen an SJS rash, those who have dealt with SJS say vigilance is key. Cases of SJS are rare in patients taking Vectibix (less than 1 in 10,000), the warning noted, but are possible.
Rather than wait for Stevens Johnson Syndrome lawsuits, and other issues to arise, Health Canada and Amgen Canada decided to add the risk of SJS/TEN to Vectibix’s warning label. It was also published to the Canadian Product Monograph. To date, no other agencies have posted a similar warning.
History of Panitumubab
Panitumubab, marketed by Amgen as Vectibix, is a monotherapy looked to after other chemotherapy regimens have failed to stop the progression of colorectal cancer. It was approved by the U.S. Food and Drug Administration (FDA) in 2006, the European Medicines Agency (EMEA) in 2007, and by Health Canada in 2008 “for the treatment of refractory EGFR-expressing metastatic colorectal cancer in patients with non-mutated (wild-type) KRAS.” An infusion solution administered through an IV, it is given to patients by doctors.
The KRAS gene performs a vital function in normal tissue signaling, and the mutation of the gene can indicate the development of many cancers. Panitumumab is “the first monoclonal antibody to exhibit the use of KRAS as a predictive biomarker.” Panitumubab is also shown to have a predilection for skin-related side effects. On the Vectibix label, it says, “WARNING: Dermatologic Toxicity…Dermatologic toxicities occurred in 90% of patients and were severe (NCI-CTC grade 3 and higher) in 15% of patients receiving Vectibix monotherapy.”
What is Stevens Johnson Syndrome (SJS)?
Stevens Johnson Syndrome, or SJS, is an autoimmune disease. Expressed as an allergic reaction to various medications, it is an extremely rare situation in which the body attacks its own skin. Initially causing a painful rash, if not treated SJS escalates to blisters, then sores, and finally ulcers. The damage can spread to mucous membranes and the eyes. Victims can suffer permanent scarring, blindness, and internal organ damage. In some cases, SJS can morph into toxic epidermal necrolysis (TEN). The next step in SJS progression, the skin reaction in TEN is similar to second- and third-degree burns and is treated in burn units. Both can prove fatal.
SJS lawsuits around the country are being filed by victims of SJS and TEN. They allege that many of the pharmaceutical companies knew the risks associated with their medication, but neglected to warn consumers.
In general, Dilantin Stevens Johnson Syndrome lawsuits are filed individually by each plaintiff and are not class actions.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Help for Victims of Stevens Johnson Syndrome from Dilantin
If you or a loved one took Dilantin and was then diagnosed with SJS or TEN, you may have a Dilantin legal claim. Discover more about what you can do at the Dilantin Stevens Johnson Syndrome (SJS) Class Action Lawsuit Investigation today.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Oops! We could not locate your form.












One thought on SJS Warning Added to Vectibix Label