 A patient who had to have her gallbladder removed after taking Benicar has filed suit against the drug’s manufacturers, alleging they failed to publish proper warnings about the drug’s side effects.
A patient who had to have her gallbladder removed after taking Benicar has filed suit against the drug’s manufacturers, alleging they failed to publish proper warnings about the drug’s side effects.
Plaintiff Doris G. of Virginia is suing drug manufacturers Daiichi Sankyo and Forest Laboratories over complications she says she suffered after taking Benicar.
She says she started taking the anti-hypertensive drug sometime in 2003.
Doris alleges she took both Benicar and a Benicar variant called Benicar HCT.
The latter combines Benicar’s active ingredient olmesartan with hydrochlorothiazide, another type of cardiovascular medication.
Plaintiff Says Gallbladder Removed After Taking Benicar
Sometime after she started taking these medications, Doris alleges, she began suffering from chronic diarrhea, vomiting and dehydration.
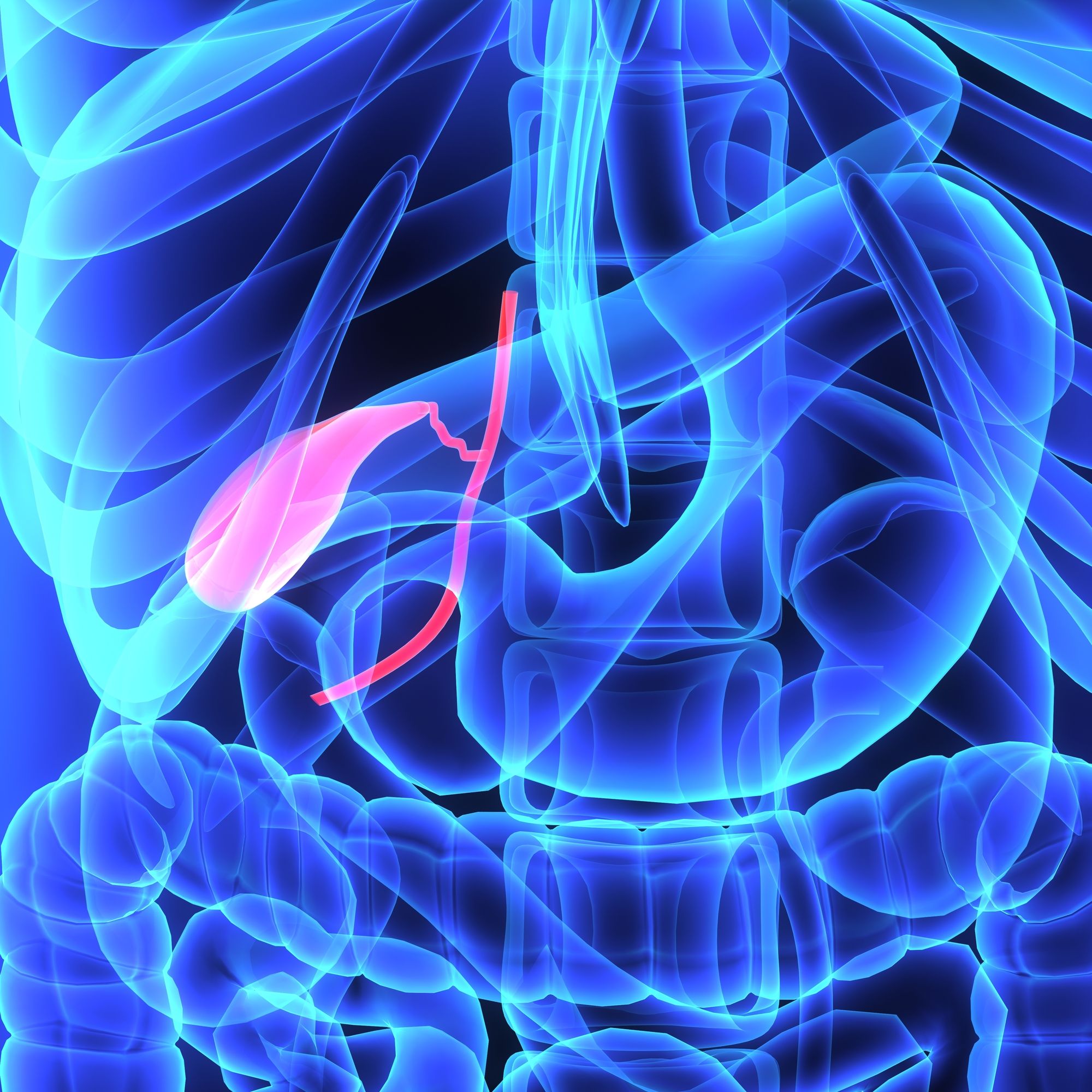
She reports losing 20 percent of her body weight. She says she suffered deficiencies of iron, potassium and vitamin B, and she eventually had to have her gallbladder removed after taking Benicar.
Doris now says these complications of having to have her gallbladder removed after taking Benicar were directly caused by the Benicar and Benicar HCT she took.
Her Benicar lawsuit seeks an award of damages, court costs, attorney’s fees, interest, and any other relief the court deems is warranted.
Benicar Lawsuits Consolidated in New Jersey
Doris’s Benicar lawsuit is one of many similar claims in a multidistrict litigation, or MDL, going on in a New Jersey federal court.
The Benicar MDL was created in April 2015 to promote more efficient pretrial procedures for the many Benicar lawsuits expected to be filed.
Consolidation in New Jersey is also expected to facilitate coordination with a few similar Benicar lawsuits also pending in New Jersey state court.
Generally, plaintiffs in these claims suffered from severe gastrointestinal symptoms – or, like Doris, had to have their gallbladder removed after taking Benicar.
This set of side effects is frequently referred to as sprue-like enteropathy, a condition whose symptoms mimic those of celiac disease.
Characteristic symptoms of sprue-like enteropathy include severe and chronic diarrhea, often accompanied by dehydration, malnutrition and weight loss.
Plaintiffs allege Daiichi Sankyo and Forest Laboratories knew about the risk of these side effects, yet failed to warn the medical community and the general public about those risks.
In 2013, the FDA issued a formal warning alerting patients and doctors to the risk of sprue-like enteropathy associated with Benicar and other olmesartan-based drugs.
The agency said it had received 23 reports of serious cases of diarrhea in association with olmesartan.
Simultaneous with that announcement, the FDA required a change to the drugs’ warning labels to include mention of the risk of sprue-like enteropathy.
In August, U.S. District Judge Robert B. Kugler backtracked his earlier decision to send a group of claims to Missouri state court.
Defendants Daiichi and Forest succeeded in persuading the judge that the Missouri plaintiffs’ claims fell within federal court jurisdiction and could therefore be properly included in the MDL in New Jersey.
Judge Kugler agreed and gave the Missouri plaintiffs 10 days to refile their claims individually.
The Benicar MDL is In re: Benicar (Olmesartan) Products Liability Litigation, Case No. 1:15-md-2606, in the U.S. District Court for the District of New Jersey.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or Benicar class action lawsuit is best for you. [In general, Benicar lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Benicar Class Action Lawsuit Investigation
If you or a loved one used Benicar for at least four months and were hospitalized for severe gastrointestinal problems, you may have a legal claim. Submit your information now for a free case evaluation.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Please Note: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client, if you qualify, or getting you dropped as a client.
Oops! We could not locate your form.












