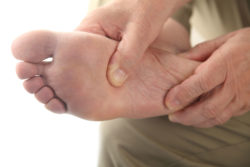
 Results from an Invokana amputation study prompted the FDA to require new warnings for the drug’s label to reflect the potential risk for leg and foot amputations.
Results from an Invokana amputation study prompted the FDA to require new warnings for the drug’s label to reflect the potential risk for leg and foot amputations.
Invokana (canagliflozin) is type-2 diabetes medication which treats the condition by filtering excess glucose out of the body through the kidneys. In May 2017, the U.S. Food and Drug Administration (FDA) released a safety announcement warning that an Invokana amputation study found that the diabetes drug significantly increased the risk of a leg or foot amputation.
“An approximately two-fold increased risk of lower limb amputations associated with canagliflozin use was observed in two large, randomized, placebo-controlled trials evaluating patients with type 2 diabetes who had either established cardiovascular disease or were at risk for cardiovascular disease,” the FDA said in their announcement.
The Invokana amputation study results were from two related studies: CANVAS (Canagliflozin Cardiovascular Assessment Study) and CANVAS-R (A Study of the Effects of Canagliflozin on Renal Endpoints in Adult Participants With Type 2 Diabetes Mellitus).
The CANVAS study showed that over one year, 5.9 out of every 1,000 Invokana-treated patients suffered from a lower limb amputation. In the placebo-treated population, 2.8 out of every 1,000 patients suffered from a lower limb amputation. The CANVAS Invokana amputation study had a significant, almost twofold increased risk for lower limb amputations.
According to the FDA safety announcement amputations of the toe and the middle of the foot were the most common. However, leg amputations were reported both above and below the knee, and some patients experienced more than one amputation.
The studies only showed the relationship between Invokana use and an increased risk for amputation. Although the exact reasons are not confirmed, some individuals argue that Invokana increases risk for amputation because it filters out glucose through the kidneys.
It is theorized that this overworks the kidneys, causing a host of circulatory and blood volume issues. These complications are associated with a decreased ability to fight infection and a weakened healing ability. When an individual is suffering from these complications, diabetic ulcers may be harder to heal and more likely to become infected, leading to amputation.
Regardless of why Invokana is associated with amputations, the statistical significance shown by Invokana amputation study results prompted the FDA to require new warnings for the drug including a boxed warning.
“We are requiring new warnings, including our most prominent Boxed Warning, to be added to the canagliflozin drug labels to describe this risk,” said the FDA regarding results from the Invokana amputation study.
Since the FDA’s safety announcement about Invokana amputation study results, numerous Invokana lawsuits have been filed against the drugs’ manufacturers. Plaintiffs in these cases often argue that manufacturers knew or should have known about the risks associated with their products but failed to warn patients.
If you or a loved one suffered from an amputation while taking Invokana, you may be eligible to take legal action against the drug’s manufacturer. An Invokana lawsuit could help recover compensation for medical expenses, pain and suffering, permanent disability, and more.
In general, Invokana and Invokamet lawsuits are filed individually by each plaintiff and are not class actions.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Invokana Class Action Lawsuit Investigation
If you or a loved one suffered ketoacidosis or lower extremity amputation after taking Invokana, Invokamet, or Invokamet XR, you may have a legal claim. See if you qualify to pursue compensation and join a free diabetes medication class action lawsuit investigation by submitting your information for a free case evaluation.
An attorney will contact you if you qualify to discuss the details of your potential case.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
Oops! We could not locate your form.












