 The U.S. Food and Drug Administration (FDA) warns that anti-seizure drugs such as Onfi (clobazam) can cause the rare but often fatal skin reactions known as Stevens Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN).
The U.S. Food and Drug Administration (FDA) warns that anti-seizure drugs such as Onfi (clobazam) can cause the rare but often fatal skin reactions known as Stevens Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN).
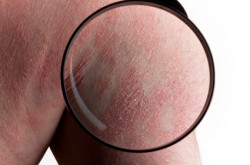
SJS symptoms are extremely painful conditions and mimic flulike symptoms – fever, fatigue and sore throat – and then manifest with lesions and ulcers forming on the mouth, lips, genital and anal regions followed by rashes covering the body. The rashes look like burns and open sores and result in skin sloughing off in large swaths. Painful blisters also develop on the mucous membranes, such as the eyes, nose, mouth and genitals. Patients may experience joint and muscle pain and a sensation that their body is burning.
Up to 40 percent of people who suffer TEN die from complications associated with the painful disease, compared with 5 to 15 percent of those with SJS. Survivors may experience lifelong health problems such as a secondary skin infection (cellulitis), sepsis (a blood infection), blindness, internal organ damage and permanent skin damage.
According to the Canadian nonprofit Milne Stevens Johnson Syndrome Society, more than 90 percent of SJS skin reactions are initially misdiagnosed as chicken pox or hand, foot and mouth disease.
More than 100,000 outpatients visit U.S. hospitals for SJS each year, while some 5,000 of those being admitted to hospitals and burn units, according to the organization. About 2 million people seek treatment for immediate hypersensitivity reactions that may be due to drugs.
FDA Reports Onfi SJS Risks
Onfi is an anti-seizure medication manufactured by Lundbeck, an international Danish pharmaceutical company that primarily focuses on the research, development and marketing of drugs to treat central nervous system disorders.
A safety announcement and label change was issued by the FDA on Dec. 3, 2013, alerting patients and medical professionals of the risk associated with Onfi, a benzodiazepine medication used in combination with other medicines to treat seizures associated with Lennox-Gastaut Syndrome, a severe form of epilepsy that causes debilitating seizures.
While Stevens Johnson Syndrome and Toxic Epidermal Necrolysis can occur at any time while taking Onfi, the dangerous skin reactions are most likely to occur during the first eight weeks, the FDA reports, or when Onfi is stopped or re-started.
In October 2011, the FDA approved Onfi for patients 2 years and older with Lennox-Gastaut Syndrome. Between that time and September 2013, some 31,000 patients received a prescription for Onfi from a U.S. retail pharmacy, according to the agency.
The FDA cautions that patients should not stop taking Onfi without first talking to their health care professionals. Doing so can result in serious withdrawal problems, such as seizures that will not stop, hallucinations, shaking, nervousness, and stomach or muscle cramps.
Those at greatest risk for Stevens Johnson Syndrome and its more deadly form, Toxic Epidermal Necrolysis, from taking Onfi are people with viral infections, a weakened immune system, a personal or family history of the disease, and carriers of a gene called HLA-B 1502, most often found in people of Chinese, Southeast Asian or Indian descent, according to the Mayo Clinic.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The Stevens Johnson Syndrome attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. [In general, SJS lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Help for Victims of Stevens Johnson Syndrome
If you or a loved one were diagnosed with Stevens Johnson Syndrome (SJS) or toxic epidermal necrolysis (TEN) after taking a prescribed or over-the-counter medication, you may be eligible to take legal action against the drug’s manufacturer. Filing an SJS lawsuit or class action lawsuit may help you obtain compensation for medical bills, pain and suffering, and other damages. Obtain a free and confidential review of your case by filling out the form below.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Oops! We could not locate your form.












