Woman Files Biomet M2a Magnum Hip Implant Lawsuit After Undergoing Two Revision Surgeries
By Sarah Pierce
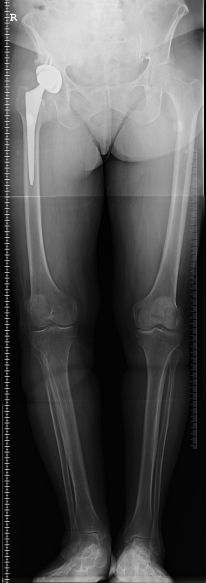 A woman who had to undergo two revision surgeries to replace her failed Biomet M2a Magnum hip implants has filed a lawsuit against the manufacturers, alleging they hid information from the public about the high failure rate of the device. According to her Biomet M2a Magnum lawsuit, over 100 adverse event reports associated with the metal-on-metal hip implant were filed at the time she received her first implant. Had the Plaintiff or her orthopedic surgeon known about these reports, they would not have used the hip replacement system, the lawsuit says.
A woman who had to undergo two revision surgeries to replace her failed Biomet M2a Magnum hip implants has filed a lawsuit against the manufacturers, alleging they hid information from the public about the high failure rate of the device. According to her Biomet M2a Magnum lawsuit, over 100 adverse event reports associated with the metal-on-metal hip implant were filed at the time she received her first implant. Had the Plaintiff or her orthopedic surgeon known about these reports, they would not have used the hip replacement system, the lawsuit says.The Biomet M2a Magnum hip system is defective, the lawsuit says, because it uses a “monoblock” system that prevents the patient from being able to use any type of acetabular liner. As a result, the metal surfaces of the all-metal hip implant rub together when the full weight of the body is applied. This defective metal-on-metal design causes an excessive amount of cobalt and chromium ions to shed off from the implant and enter into the bloodstream. When this happens, the surrounding tissue becomes inflamed, causing pain and local tissue and bone deterioration. Patients implanted with the defective metal-on-metal hip implant will begin to notice squeaking and popping sounds, swelling, looseness and dislocation when this occurs.
Updated August 7th, 2012
{loadposition content_postads}
All medical device, dangerous drug and medical class action and lawsuit news updates are listed in the Drug and Medical Device section of Top Class Actions.
Top Class Actions Legal Statement














