New Study Reveals Metal-on-Metal Hip Implant Risks
By Christina Drury
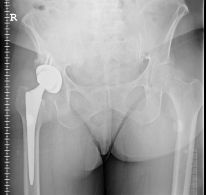 Metal-on-metal implant lawsuits continue to make their way through the court system as new studies report new risk factors. On December 18, 2012, “Arthritis Care & Research” reported that according to the FDA, patients who are 75 years old or younger at the time of their first hip replacement surgery are at a higher risk for revisions than those older than 75. Patients who are heavier or taller are also at risk for higher rates of metal hip implant failure and revision surgery, as well as patients who have had previous orthopedic surgery or have received a cemented femoral component.
Metal-on-metal implant lawsuits continue to make their way through the court system as new studies report new risk factors. On December 18, 2012, “Arthritis Care & Research” reported that according to the FDA, patients who are 75 years old or younger at the time of their first hip replacement surgery are at a higher risk for revisions than those older than 75. Patients who are heavier or taller are also at risk for higher rates of metal hip implant failure and revision surgery, as well as patients who have had previous orthopedic surgery or have received a cemented femoral component. First U.S. Study of Hip Replacement Risks
Conducted at Harvard Medical School and Brigham and Women’s Hospital, this was the United States’ first study on the risk factors related to those who underwent hip replacement surgeries. The team that conducted the study reviewed a variety of data sources on 719 patients, including their hospital records. All of the records that were examined belonged to patients who had undergone a hip replacement surgery between July of 1995 and June of 1996. Each patient was paired with a similar control patient and tracked for 12 years. A nationwide law firm has cautioned that those who have undergone or will undergo hip replacement surgery should be aware of the risk factors of metal-on-metal hip implants as this type is generally more geared towards younger patients.
Current Lawsuits against Metal-on-Metal Hip Implants
Several metal-on-metal hip implant manufacturers have been included in a number of lawsuits that have been brought against the medical device. The lawsuits allege the metal-on-metal hip implant is predisposed to premature failure and patients who have these devices implanted are at risk of a dangerous exposure to high amount of metal ions. Lawsuits are being actively filed in a multidistrict litigation that has been established in the U.S. District Court, Northern District of Ohio. These claims involve the DePuy ASR hip implant device. Additional lawsuits are also being filed in the Northern District of Texas. These lawsuits concern metal-on-metal DePuy Pinnacle hip devices.
FDA Orders Post-Market Surveillance
The FDA issued an order to have the metal-on-metal hip implant devices closely monitored. The order was given to the manufacturers to study the effects of the devices in May 2011. The FDA intended to research claims that metal particles that may be shed by the metal-on-metal hip replacements could results in a harmful tissue reaction. This reaction has the potential to lead to bone and/or surrounding tissue damage around the joint and device. There have also been reports that some patients who have had high levels of metal ions in their bloodstream have suffered various other symptoms and illnesses in other areas of their body, including the heart, thyroid gland and nervous system.
Know the Facts, Know Your Rights
If you or a loved one has undergone hip replacement surgery to receive a metal-on-metal hip implant and has suffered from any injuries as a result, you may be entitled to compensation. Visit our DePuy, Biomet, Wright Medical Technology & Others, Metal on Metal Hip Implant Class Action Lawsuit Investigation page. Learn more about the lawsuit and how you can get involved.
Updated January 1st, 2013
All medical device, dangerous drug and medical class action and lawsuit news updates are listed in the Drug and Medical Device section of Top Class Actions.
Top Class Actions Legal Statement