Top Class Actions’s website and social media posts use affiliate links. If you make a purchase using such links, we may receive a commission, but it will not result in any additional charges to you. Please review our Affiliate Link Disclosure for more information.
Several patients report experiences with Injectafer that prompted them to warn others to be wary of the iron-boosting medication. Injectafer side effects can range from irritating to debilitating.
What is Anemia?
A person who is anemic suffers from low levels of iron in the blood. Every person’s blood needs iron to produce hemoglobin, which is the portion of red blood cells that distribute oxygen throughout the body.
Women are at a greater risk of anemia, and symptoms may include:
- Weakness
- Paleness
- Short of breath
- Extreme fatigue
- Dizziness
- Cold extremities
- Brittle nails
- Sore tongue
- Strange cravings for ice or dirt
Many times, doctors will recommend patients take iron supplements orally, but when that fails to sufficiently boost blood iron levels, a prescription infusion medication is sometimes ordered.
What is Injectafer and Who Makes This Drug?
People who have severe anemia or non-dialysis dependent chronic kidney disease may be prescribed Injectafer when oral iron supplements fail to help them.
A type of elemental iron, ferric carboxymaltose (FXM) is the active ingredient in Injectafer, a prescription-only medication made available by Luitpold Pharmaceuticals, Inc.; American Regent, Inc.; Daaichi Sankyo; and Vifor Pharmaceuticals Management Ltd.
Injectafer was approved by the U.S. Food and Drug Administration (FDA) in July 2013.
How is Injectafer Administered?
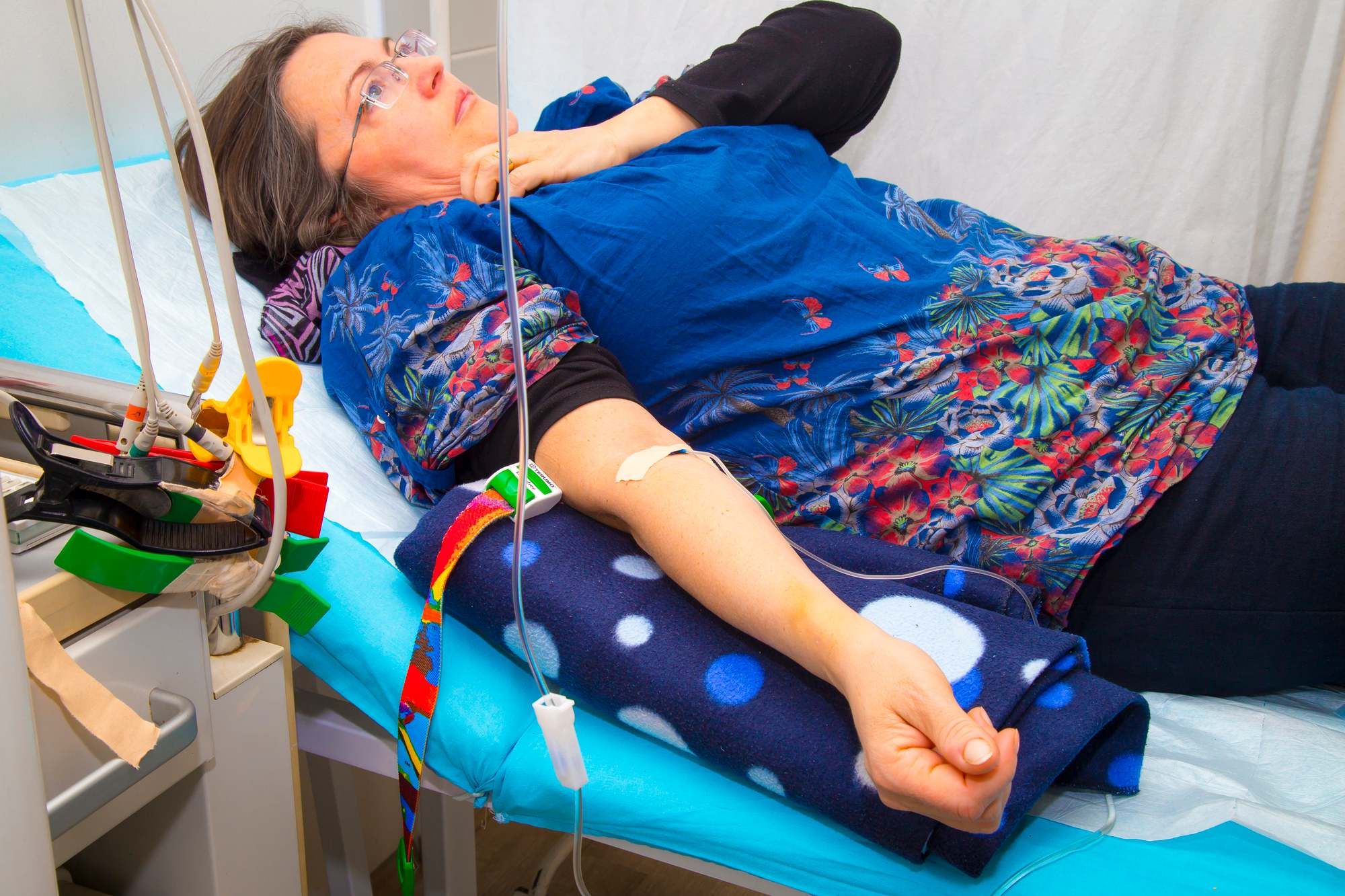
Injectafer can be provided as a slow undiluted intravenous push at a rate of about 100 mg per minute. If you or your health care provider prefer to use the slow drip infusion method, Injectafer is mixed with a sodium chloride solution that gradually enters the blood stream directly through a vein.
No matter which method is used, the health care provider will ask you to stay an additional half hour after the drug is administered to ensure you do not have an allergic reaction.
Injectafer is normally given in two doses at least seven days apart.
What Do Injectafer Reviews Say?
Patients have reported suffering from a potentially fatal condition called hypophosphatemia, which means the blood’s phosphorous levels are dangerously low.
In December of last year, one patient said three months after receiving a second round of Injectafer, her “muscles were like wet noodles.”
She said she very suddenly experienced an Injectafer delayed reaction when she began to have trouble walking and suffered severe weakness and cramping in her muscles, especially those in her legs. She said her doctors were not aware that Injectafer could cause a dangerously low phosphorous count, which meant she had to show them study results and law suits in order to convince her doctors to conduct a blood test on her phosphorous levels. She believes her own research very likely saved her life because that blood work proved she had severe hypophosphatemia.
Another patient said she had received infusions of other brands such as Feraheme and Venofer for a decade without having any problems. Her doctor switched her to Injectafer, but after the first dose, she said she began suffering from a severe case of hypophosphatemia that required her to be hospitalized twice in an effort to increase her phosphorous levels. She said the phosphorous infusions have not yet helped. In the meantime, she alleges she gained eight pounds in five days due to inflammation, is extremely fatigued and fighting severe muscle pains. She wrote, “This is a nightmare. Do NOT USE THIS DRUG!”
More Bad Experiences with Injectafer
A patient going by the name of Ro said she found just two Injectafer infusions left her with severe muscle spasms, weakness and peripheral neuropathy. She said she ended up in the emergency room due to hypophosphatemia, which also caused her bones to weaken.
Is Injectafer Safe?
The research noted that in a second trial of patients who received a competing medication call Monofer had much different results. Only 8.1 of patients who received Monofer experienced hypophosphatemia, and none were diagnosed with a severely low phosphorous levels.
Hypophosphatemia has been linked with a number of possibly fatal health problems, including:
- Respiratory failure
- Cardiac arrest
- Irregular heart rhythm
- Destruction of red blood cells
- Seizures
- Muscle weakness or death of muscle tissue
- Mental confusion or delirium
- Comatose state
- Death
Should I File an Injectafer Lawsuit?
If your experiences with Injectafer include serious injuries such as hypophosphatemia, you could be eligible to participate in this class action lawsuit investigation.
Financial compensation may be available to you for lost wages, physical pain, mental suffering, future medical needs and other related losses.
An experienced Injectafer attorney can help you navigate the legal avenues required to ensure you receive the optimal award possible.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2024 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Free Injectafer Harmful Side Effects Lawsuit Evaluation
Fill out the form below for a free case evaluation. If you qualify, a lawyer will contact you to discuss the details of your potential case at no charge to you.