Virginia plaintiff India W. is suing pharmaceutical giants Bayer Pharmaceuticals and Janssen Pharmaceuticals, a Johnson & Johnson subsidiary, for allegedly causing her to suffer near-fatal bleeding injuries. India claims that in direct result of using the defendants’ product, Xarelto, she suffered internal bleeding, and still remains under her physician’s watch.
Like millions of patients across the United States, India had been suffering from atrial fibrillation and had needed to be prescribed a blood-thinner. After reviewing the defendant’s advertisements, the plaintiff’s physician prescribed her Xarelto on April 22, 2013. According to the Xarelto lawsuit, India had followed all prescription instructions, as well as the advice of her physician.
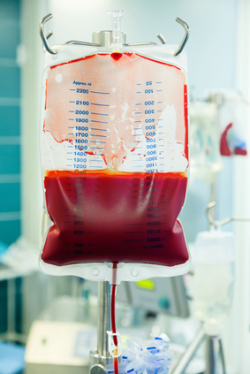
Despite this diligence, India started suffering side effects less than a month after starting Xarelto, reporting severe fatigue and black stool to her physician on May 5, 2013. On that day, India was rushed to the emergency room and admitted to the intensive care unit where she was administered an endoscopy. This diagnostic test revealed she was suffering from gastrointestinal bleeding. Additional blood tests showed that her hemoglobin levels were very low and required a blood transfusion of one gallon.
After the transfusion, India was immediately taken off Xarelto and stayed in the hospital for several days, receiving multiple blood transfusions. During her stay, she reportedly was given a total of four units of blood. India also had to be treated with iron sulfate due to an iron deficiency anemia she developed due to her gastrointestinal intestinal bleeding event. Even though she survived her internal bleeding event, India and her physician still remain alert for any lasting side effects of Xarelto.
At no point before or after her Xarelto prescription did the defendant inform the plaintiff of the potential bleeding side effects associated with their anticoagulant, the Xarelto lawsuit alleges. There were no warnings or indications on Xarelto’s instruction label, nor did the defendant provide any kind of information on any relevant studies.
India is filing legal action against Janssen and Bayer Pharmaceuticals for failing to protect her against the drug’s side effects. The plaintiff’s lawyers state that the drug companies had the civil responsibility of warning their client and other Xarelto patients of the possible dangers their drug comes with, because they rely on the given information. India insists that she never would have taken Xarelto if she had known about the potential risk of gastrointestinal bleeding.
India is suing Janssen Pharmaceuticals and Bayer Pharmaceuticals for being allegedly responsible for manufacturing, selling, and marketing a dangerous drug. The charges include negligence, false advertising, concealing information, and misrepresenting a product.
This Xarelto Lawsuit is Case No. 2:15-cv-01492-EEF-MBN, in the U.S. District Court for the Eastern District of Louisiana.
Overview of Xarelto Bleeding Complications
Xarelto is the second in the line of successors for Warfarin, with many pharmaceutical experts calling them the new-generation anticoagulants. For years, Warfarin had been the only blood-thinner available for patients suffering from atrial fibrillation or patients suffering blood-clotting after undergoing knee replacement surgery.
Now, there are several other options patients can choose from to treat these conditions, including Xarelto. This drug was approved by the FDA in 2011, and was released as a joint effort between Johnson & Johnson and Bayer Pharmaceuticals, to attempt to break into the lucrative anticoagulant market.
Through aggressive sales and marketing, the investment proved to be a success as Xarelto dethroned Pradaxa, approved only a year prior, as the best-selling anticoagulant in the United States. Unfortunately, it was discovered that Xarelto had the same severe bleeding side effects as Pradaxa, and had the same design flaws as the earlier blood-thinner as well. While the new-generation anticoagulants are advertised to be more efficient and fast-acting than Warfarin, they lack two important safety features of the original blood-thinner.
Xarelto and the other new-generation blood-thinners lack an approved bleeding antidote, and do not require patients to undergo frequent dose adjustments. Experts believe that if patients had been required to frequently see their doctors, then numerous bleeding injuries could have been avoided.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The Xarelto attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or Xarelto class action lawsuit is best for you. [In general, Xarelto lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
Get Help – It’s Free
Join a Free Xarelto Class Action Lawsuit Investigation
If you or a loved one took Xarelto (rivaroxaban) and suffered injuries such as uncontrollable internal bleeding, gastrointestinal bleeding, hemorrhaging, deep vein thrombosis or pulmonary embolism, you may have a legal claim. See if you qualify by filling out the short form below.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Oops! We could not locate your form.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.












