 The Stryker V40 femoral head recall has affected thousands of metal hip implants that were recalled from the market due to reports of serious injuries and device failure.
The Stryker V40 femoral head recall has affected thousands of metal hip implants that were recalled from the market due to reports of serious injuries and device failure.
Stryker had sent notice of the Stryker V40 femoral head recall to healthcare facilities in August 2016, stating that there were failure reports of the taper locks failing in specific sizes and models of the LFIT V40 femoral heads.
The Stryker V40 femoral head recall effects parts manufactured before 2011, with the specific sizes of Stryker LFIT V40 femoral heads ranging between 36 to 44 millimeters and offsets between +4 to +12. Since the Stryker V40 femoral head recall was initiated, healthcare facilities have purged the affected medical devices from devices from their inventory to prevent future incidents.
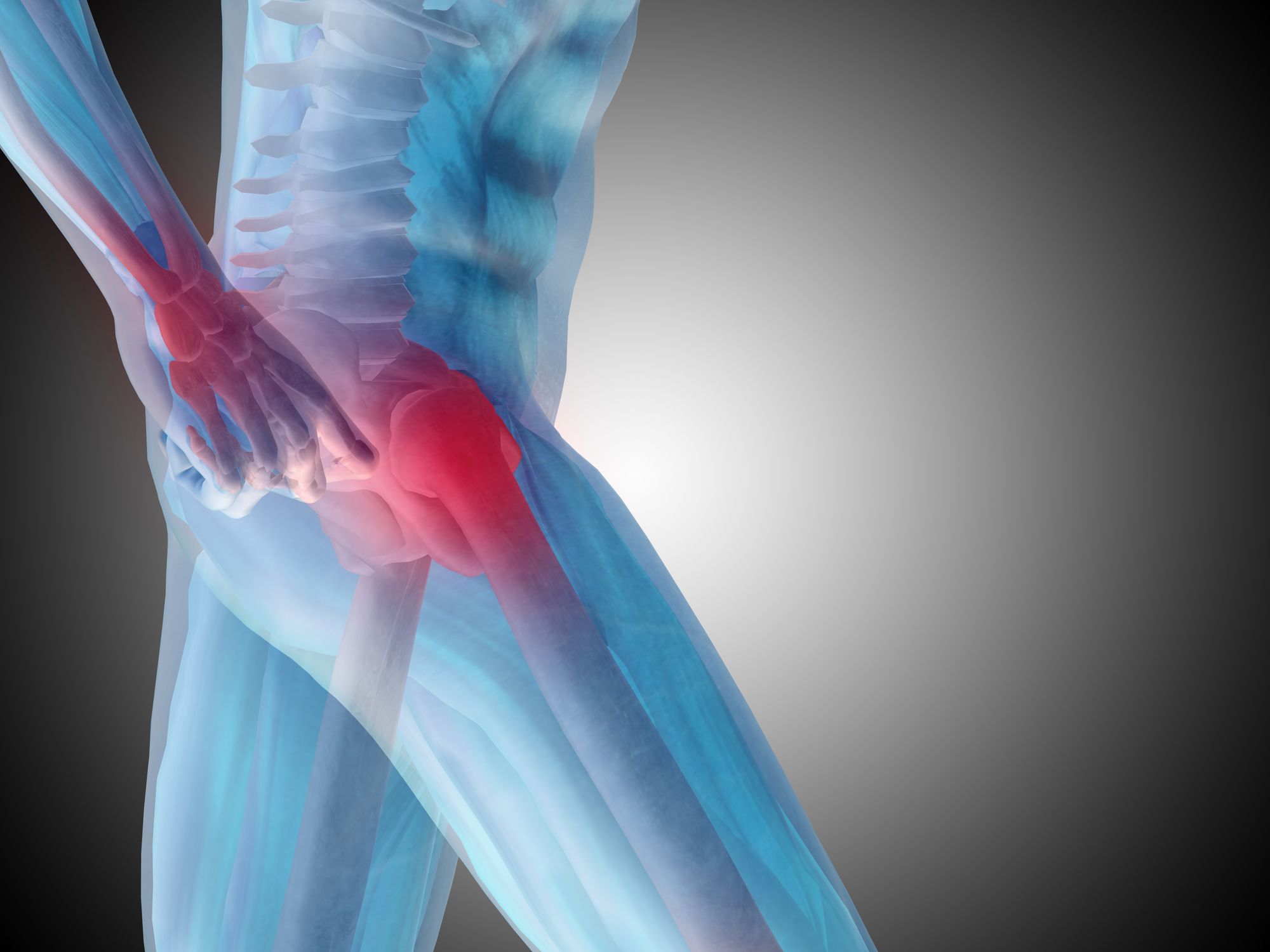
These V40 femoral heads are parts of the metal hip implants, which are implanted at the upper end of the femur bone while the other end of the femoral heads are directly implanted inside the pelvic bone.
Stryker metal hip implants are typically prescribed to patients who have sustained damage to their hip bones, and these patients often suffer from limited movement and pain. The Stryker V40 femoral head recall was initiated after the company had received a higher number of injury reports than expected.
Overview of Metal Hip Implant Complications
The failure reports indicate the femoral heads may disconnect from the rest of the implant, which could weaken the overall structure of the device and surrounding bone potentially resulting in hip stem trunnion fracture and metal debris being shed into the bloodstream and surrounding tissue. Different parts of the device failing could lead to the metal hip implant not working at all, which will ultimately compound the patients’ condition.
This is not the first time Stryker has conducted a market recall of their metal on metal hip implant systems, as the company issued a voluntary recall of its Rejuvenate and ABG II metal hip implant systems in July 2012.
Stryker had reportedly issued this recall due to reports of serious metal hip implant complications such as metallosis and device failure. Metallosis is the condition in which metal ion levels in the blood raise to dangerous levels which are caused by the metal debris shed by the Stryker metal hip implants.
When the ball and socket joints of the metal on metal hip implants interact, metal ions are shed into the body and surrounding tissue. This complication may lead to device failure, tissue death, corrosion, fretting, and formation of pseudotumros. Oftentimes these metal hip implant complications force patients to undergo revision surgery to resolve these problems.
It is important to note that revision surgery is considered riskier than the initial implant surgery, because the risks of complications are higher. Many patients allege Stryker may have known about these risks before the Stryker V40 femoral head recall was initiated and failed to warn them.
Patients who experienced injuries from the Stryker V40 femoral head component may be able to file legal action against the manufacturer. Potential claimants should contact a lawyer, to determine eligibility for a Stryker V40 femoral head lawsuit.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The hip implant attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. [In general, metal hip implant lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Stryker Metal Hip Replacement Class Action Lawsuit Investigation
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Please Note: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client, if you qualify, or getting you dropped as a client.
Email any problems with this form to [email protected].
Oops! We could not locate your form.












