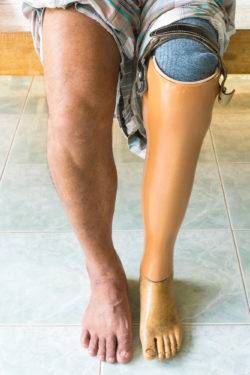
 In a new lawsuit filed in U.S. District Court for the District of New Jersey on June 5, 2018, the plaintiff alleges that he became a leg amputee as a direct consequence of taking the diabetes drug Invokana.
In a new lawsuit filed in U.S. District Court for the District of New Jersey on June 5, 2018, the plaintiff alleges that he became a leg amputee as a direct consequence of taking the diabetes drug Invokana.
Plaintiff Ronnie F. seeks to hold Janssen Pharmaceuticals Inc., Janssen Research and Development LLC as well as the parent company Johnson & Johnson Inc. responsible for the injuries he claims came from his use of Invokana.
According to the lawsuit narrative, the diabetes drug Invokana was the first in its medication class to get market approval from the U.S. Food and Drug Administration (FDA).
This commercial release of the drug for the treatment of type-2 diabetes, or adult-onset diabetes, came in March 2013. The diabetes drug Invokana is also known generically as canagliflozin and is a sodium glucose cotransporter 2 (SGLT2) inhibitor.
What is an SGLT2 Inhibitor?
An SGLT2 inhibitor discourages renal system re-absorption of blood glucose so that excess sugars are eliminated through the body’s urine output rather than remain in the blood stream. The goal of using them is to obtain optimum blood sugar control in persons with type-2 diabetes.
This process, however, may put strain on the kidneys in people who already have risk factors secondary to the disease for renal problems and failure.
In order for the diabetes drug Invokana or any SGLT2 inhibitor to remove extra sugars through the kidneys, these organs must bind the glucose to a water molecule, according to the factual section of the lawsuit.
The resultant overall dehydration may lead to with a host of other potential problems. Reduced blood volume is a consequence of less water in the body. Blood plasma which carries platelets and white blood cells to extremities is less. These elements are necessary to kill infections and bring healing to wounds.
According to Ronnie, a patient taking the diabetes drug Invokana can easily have a small leg wound turn infectious and not be able to fight it off. Once it spreads, it can involve the nearest bone and allow for gangrene to set in. In this situation, the only safe solution is for the patient to become a leg amputee to keep the infection from turning systemic.
Other Dangers of the Diabetes Drug Invokana
Another danger of Invokana and other SGLT2’s is ketoacidosis—a condition in which the body relies upon fat for primary fuel instead of glucose. A byproduct of this process is excessive blood acids known as ketones.
Ketoacidosis generally comes with excessive blood glucose levels and frequent trips to the bathroom to urinate due to strain on the kidneys. This is an emergency body-fueling process which the diabetes drug Invokana may put the body into, according to the factual section of the lawsuit.
FDA Warnings, Label Changes, and the Plaintiff’s Story
Beginning in late 2015, the FDA began to require label changes on Invokana. The defendants were required to warn of mild and fatal ketoacidosis, kidney injury and failure, and increased risk of needed amputation. A study published in 2017 warned patients of the increased risk of becoming a toe, foot, or leg amputee with this medication.
Ronnie says he was prescribed Invokana beginning in March 2017. By August of that same year, he says he had to have his right leg amputated. He allegedly required extensive hospitalization and treatment to recover. He is seeking compensatory and punitive damages against the defendants.
The Diabetes Drug Invokana Lawsuit is Case No. 3:18-cv-10205-BRM-LHG, in the U.S. District Court for the District of New Jersey.
In general, Invokana and Invokamet lawsuits are filed individually by each plaintiff and are not class actions.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Invokana Class Action Lawsuit Investigation
If you or a loved one suffered ketoacidosis or lower extremity amputation after taking Invokana, Invokamet, or Invokamet XR, you may have a legal claim. See if you qualify to pursue compensation and join a free diabetes medication class action lawsuit investigation by submitting your information for a free case evaluation.
An attorney will contact you if you qualify to discuss the details of your potential case.
PLEASE NOTE: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client or getting you dropped as a client.
Oops! We could not locate your form.












