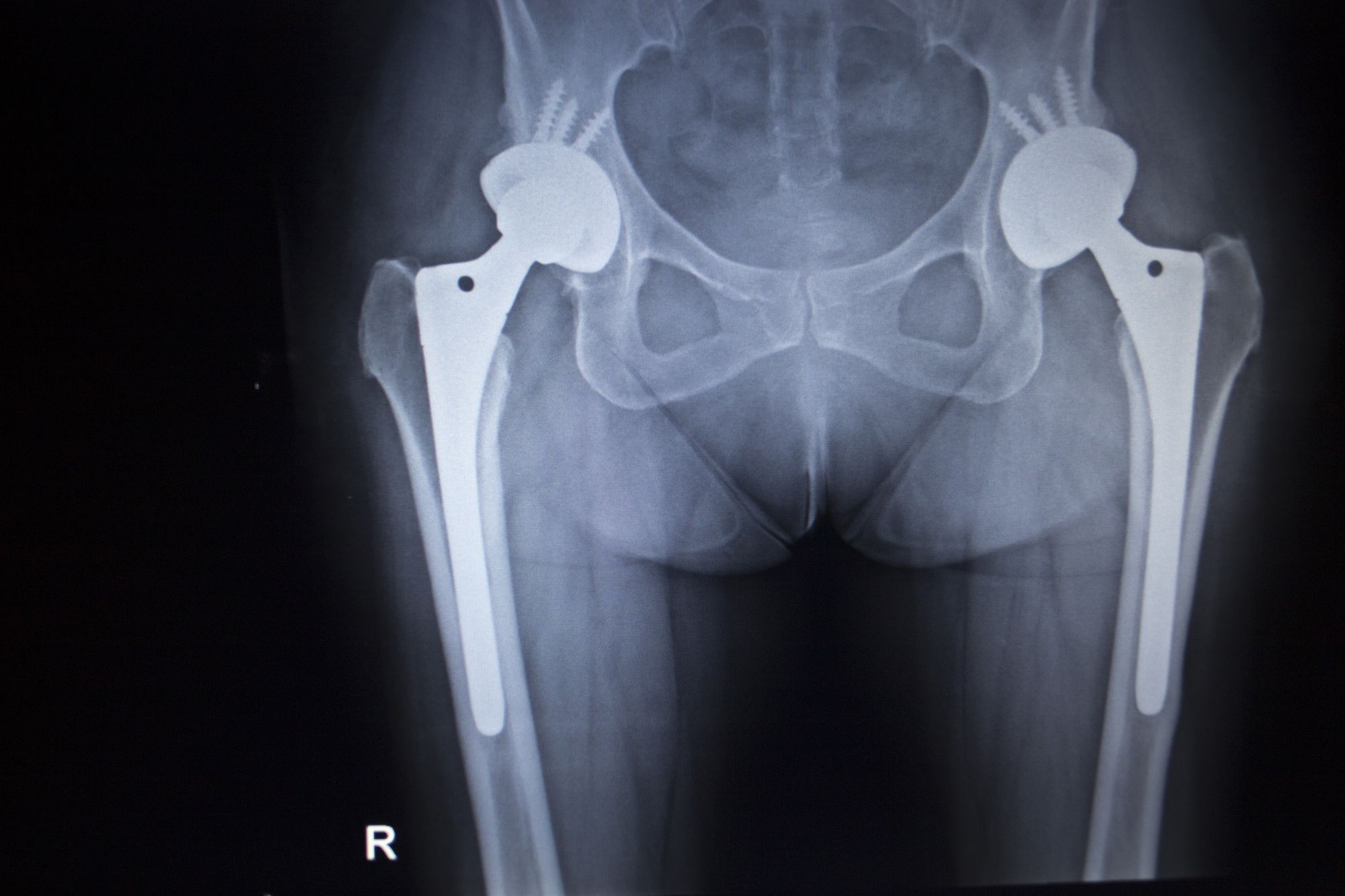
 Prosthetic manufacturing giant Stryker Corporation has issued a voluntary recall of its LFIT metal hip implant after numerous complaints were submitted.
Prosthetic manufacturing giant Stryker Corporation has issued a voluntary recall of its LFIT metal hip implant after numerous complaints were submitted.
The complaints alleged the all metal V40 femoral heads had caused numerous problems, ultimately resulting in the Stryker recall.
The complications that incited the Stryker recall included corrosion, debilitating pain, and blood metal poisoning or metallosis.
Oftentimes, these problems forced patients to undergo revision surgery to either replace or explant the metal-on-metal hip implant.
This Stryker recall affects seven different sizes of the LFIT V40 metal hip implants, with diameters ranging from 36 to 44 millimeters.
While this is not the first time Stryker Corporation has faced criticism for their all metal hip implants, this recall is the most recent in a series of similar complications reported in earlier recalls.
Like with previous metal hip implant recalls, the LFIT V40 hip implant had allegedly been shedding metal ions into the bloodstream at the ball and socket joint.
Also known as fretting, metal ions are shed into the bloodstream when the ball and socket joint components interact. This raises blood metal ion levels to dangerous heights, which causes tissue death and can negatively impact surrounding bone. This leads to further complications like pseudotumor formation, device failure, and infection.
The Stryker recall was also initiated by reports of the LFIT V40 metal hips suddenly dislocating or disconnecting, further adding to the concern surrounding the devices.
Stryker Recall Metal Hip Implant Complications
When metal hip implants were first released into the United States market, they were thought to be overtly superior to their ceramic or plastic counterparts. Metal on metal hip implants were marketed to be more durable and longer lasting, with most models being customizable to patients.
However it was not long after the first models were released that patients reported problems relating to device failure. According to a recent study conducted in July 2012 and published in the Orthopedics medical journal, approximately 95% of patients who receive all-metal hip implants had to undergo revision surgery within three years.
The numerous complaints indicated the LFIT V40 metal hip implant had similar results, which ultimately contributed to the latest Stryker recall. Previously, there had been a Stryker recall in 2012, when the company recalled the Rejuvenate and ABG II modular hip replacements due to reports of device failure and corrosion.
Similar to the LFIT V40 metal hip implants, patients who received the metal hip implant had very high levels of cobalt and chromium in their blood. Even though Stryker allegedly knew of these metal hip implant complications, the company failed to warn patients and surgeons of these problems.
Patients who have suffered metal hip implant complications from a Stryker metal hip implant may be eligible to file legal action against the company.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The hip implant attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. [In general, metal hip implant lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Stryker Metal Hip Replacement Class Action Lawsuit Investigation
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Please Note: If you want to participate in this investigation, it is imperative that you reply to the law firm if they call or email you. Failing to do so may result in you not getting signed up as a client, if you qualify, or getting you dropped as a client.
Email any problems with this form to [email protected].
Oops! We could not locate your form.












