 As far back as 2008, the FDA has indicated that components of the Infuse Bone Graft System may carry substantial risks.
As far back as 2008, the FDA has indicated that components of the Infuse Bone Graft System may carry substantial risks.
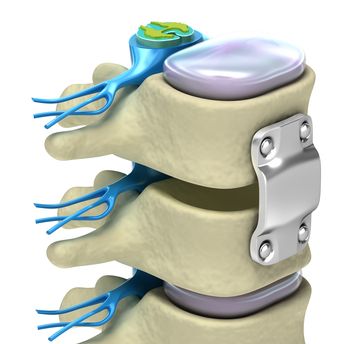
The Infuse Bone Graft System is a type of spinal implant designed to improve spinal fusion surgery. In spinal fusion surgery, surgeons join two adjacent vertebrae together into a single bone. Surgeons typically perform spinal fusion surgery to compensate for a damaged disc, a piece of connective tissue that supports individual bones in the back, which can wear out from age, disease, or injury.
In a typical spinal fusion surgery, surgeons harvest redundant bone from elsewhere in the body to encourage the bones to fuse together. The Infuse Bone Graft System is designed to streamline this process.
The Infuse Bone Graft System includes two components, a metal cage and a bioengineered gel. This gel is mixed with cells from the patient’s own body. In spinal fusion surgeries with the Infuse Bone Graft System, the metal cage holds the vertebrae in place while the bioengineered gel encourages the bone to grow. The metal cage remains in place after the bones grow together, further strengthening the fused vertebrae.
The bioengineered gel contained in the Infuse Bone Graft System is designed to streamline spinal fusion surgery by allowing surgeons to skip the separate procedure of harvesting bone from elsewhere in the body. However, Infuse Bone Graft lawsuits have alleged that the Infuse Bone Graft System may carry its own risk, and statements by government regulators may support these assertions.
As early as 2008, the US Food and Drug Administration voiced concerns about the safety of the Infuse Bone Graft System. In a Public Health Notice dated to July of 2008, the FDA stated that they had received at least 38 reports of swelling of the neck and throat and neurological problems associated with the bioengineered gel used in the Infuse Bone Graft System. In many of these cases, patients needed emergency surgery to keep them breathing as the swelling obstructed their airways.
The FDA’s statement also included warning about “off-label” use of the Infuse Bone Graft System, and stated that the device was not approved for use in the cervical vertebrae, that is, the bones of the neck.
Additional concerns have emerged subsequent to the FDA’s statement. Uncontrolled bone growth has been alleged to occur in patients who used the Infuse Bone Graft System. Various Infuse lawsuits have held that the makers of the medical implant were aware of these risks, but marketed the device anyway. Additionally, it is alleged that Medtronic Inc., the manufacturer of Infuse, aggressively promoted the device, going as far as to advocate the device in surgeries other than those approved by the FDA, termed “off-label” usage.
Under US law, it is forbidden for medical companies to promote their wares for off-label usage; companies are required to thoroughly test drugs and medical device for every purpose they intend to promote their device. In the case of the Infuse Bone Graft System, it is alleged that Medtronic Inc. engaged in a clandestine campaign of promoting off-label use through backchannels, like hiring researchers to publish fraudulent research papers promoting the Infuse Bone Graft System for off-label use.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. [In general, Infuse bone graft lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Medtronic Infuse Class Action Lawsuit Investigation
An investigation has been launched to find spinal surgery patients who were implanted with Medtronic’s Infuse bone graft and suffered complications such as nerve damage; excessive bone growth; chronic pain; difficulty breathing, swallowing, and speaking; male sterility and other uro-genital injuries. See if you qualify to take legal action by filling out the short form below.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Oops! We could not locate your form.












