 The U.S. Food and Drug Administration (FDA) has issued a warning that Onfi can cause Stevens Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN) at any time when taking this epilepsy treatment. These serious skin reactions are rare, but can cause permanent harm to patients and may even lead to death.
The U.S. Food and Drug Administration (FDA) has issued a warning that Onfi can cause Stevens Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN) at any time when taking this epilepsy treatment. These serious skin reactions are rare, but can cause permanent harm to patients and may even lead to death.
Onfi is a benzodiazepine drug that is used with other medicines to treat seizures associated with a severe form of epilepsy called Lennox-Gastaut Syndrome as well as mental disorders.
Manufactured by the Danish pharmaceutical company Lundbeck, Onfi (clobazam) was approved by the FDA in October 2011 for the treatment of patients 2 years or older suffering from Lennox-Gastaut Syndrome (LGS), a severe form of epilepsy. Since that time, about 31,000 patients have been prescribed the epilepsy drug in the United States.
Additionally, the FDA has become aware of at least 30 reported cases of SJS worldwide, including six cases in the United States. Five of the six cases in this country involved young children. At least one death and one case of blindness due to Onfi SJS have also been reported.
The Onfi Stevens Johnson Syndrome risk appears to be highest during the first eight weeks of treatment, and after a consumer has stopped and restarted the medication.
According to proposed Onfi SJS lawsuits, Lundbeck allegedly failed to adequately research the potential side effects of Onfi or warn about the Onfi SJS risk to consumers and prescribing physicians.
FDA Safety Warning for Onfi Skin Reactions
In a recent drug safety communication, the FDA warned that the anti-seizure drug Onfi can cause rare but serious skin reactions that can result in permanent harm and death. The Drug Safety Communication came after a review of adverse events linking Onfi to 20 skin reactions involving Stevens Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN). The FDA is advising clinicians to closely monitor patients taking Onfi for SJS and TEN, especially at the start of therapy or when the drug is reintroduced.
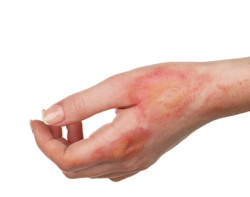
Steven Johnsons Syndrome and Toxic Epidermal Necrolysis
Stevens Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN) are rare but severe, life-threatening skin reactions that cause the top layer of skin to detach and fall off in large sheets. Sloughing skin may appear burned, while blisters may also develop on mucous membranes located at the eyes, mouth, and genitals. The Onfi SJS burns may ultimately spread from all over the body to inside the intestines, lungs, and stomach.
Onfi skin reactions are extremely dangerous, painful, disfiguring, and can lead not only to life-threatening infections, but also organ damage, blindness, and death. Common symptoms include, but are not limited to:
- Pain of the Skin
- Red or Purple Rashes that Spread from the Face
- Swelling of the Face or Tongue
- Hives
- Skins Detaching or Sloughing Off in Sheets
- Blisters on the Mouth, Nose, or Eyes
- Hives
- Tiredness
- Fever, Chills, Headaches, Muscle Aches, Joint Pains
- Sore Throat
Onfi SJS Lawsuits
Several Onfi lawsuits accuse Lundbeck of off-label promotion of their anti-seizure drug, failure to warn, defective design and manufacturing, negligence and illegally promoting Onfi for uses that were not approved by the FDA. While many Onfi users were prescribed the drug for Lennox-Gastaut Syndrome (LGS), Lundbeck allegedly marketed Onfi for a much broader range of seizure treatments. Additionally, Lundbeck Pharmaceuticals is accused of knowing about potential Onfi side effects for years and withholding information about these Onfi side effects.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The Stevens Johnson Syndrome attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or class action lawsuit is best for you. [In general, SJS lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Help for Victims of Onfi Stevens Johnson Syndrome
If you or a loved one were diagnosed with Stevens Johnson Syndrome (SJS) or toxic epidermal necrolysis (TEN) after taking Onfi, you may be eligible to take legal action against the drug’s manufacturer. Filing an Onfi SJS lawsuit or Onfi class action lawsuit may help you obtain compensation for medical bills, pain and suffering, and other damages. Obtain a free and confidential review of your case by filling out the form below.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Oops! We could not locate your form.












