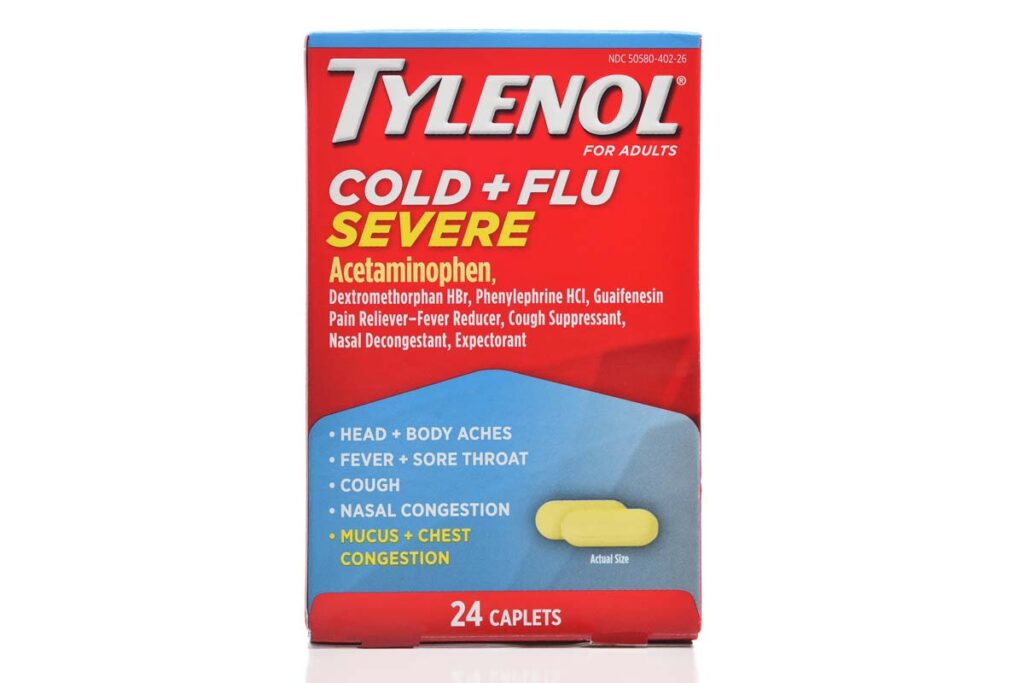
Tylenol lawsuit overview:
- Who: Kenvue Inc., McNeil Consumer Healthcare and Reckitt Benckiser are facing a class action lawsuit related to their Tylenol Cold and Flu Severe and Mucinex Sinus Max products.
- Why: The over-the-counter medications, which use phenylephrine as their active ingredient, were recently recommended to be classified as ineffective by a U.S. Food and Drug Administration advisory committee.
- Where: The Tylenol lawsuit was filed in federal court in New York.
The makers of Tylenol Cold and Flu Severe and Mucinex Sinus Max are facing a class action lawsuit related to the ineffectiveness of the active ingredient phenylephrine in their over-the-counter products.
An advisory committee for the U.S. Food and Drug Administration recently recommended that the FDA move to remove oral phenylephrine drugs used to combat nasal decongestion from the department’s list of safe and effective drugs.
Consumers purchased the drugs believing they would be effective when the companies that made those drugs – Kenvue Inc., McNeil Consumer Healthcare and Reckitt Benckiser – should have known that they were not effective, the Mucinex lawsuit claims.
“Unfortunately for plaintiff and the putative class members, defendants willfully ignored scientific and industry knowledge concerning the lack of effectiveness of PE Drugs for treating the indications identified, and performed inadequate testing and quality oversight of the PE Drugs ascertain the true efficacy of PE Drugs for treating the indications identified (mainly, nasal congestion),” said the class action filed by Plaintiff Chioma Ozuzu.
Manufacturers should have known phenylephrine OTC drugs were ineffective and stopped selling them, lawsuit claims
The manufacturers should pay customers back for the full purchase price of the phenylephrine drugs, the Mucinex lawsuit says, along with requesting an injunction against further sale of the products.
The companies knew that the products were not effective from previous studies on phenylephrine but have continued to sell and advertise the products as being effective, the lawsuit claims.
Sale of the Tylenol and Mucinex phenylephrine over-the-counter drugs violates express warranties, implied warranties, the Magnuson-Moss Warranty Act and falls under the category of fraud due to the misrepresentations about the drug, the Tylenol lawsuit claims.
Have you purchased Tylenol Cold and Flu Severe or Mucinex Sinus Max? Let us know in the comments.
The plaintiff is represented by Raymond S. Silverman, Melanie H. Muhlstock and Jason S. Goldstein of Parker Waichman LLP.
The Tylenol class action lawsuit is Ozuzu v. Kenvue Inc., et al., Case No. 1:23-cv-07395, in the U.S. District Court for the Eastern District of New York.
Don’t Miss Out!
Check out our list of Class Action Lawsuits and Class Action Settlements you may qualify to join!
Read About More Class Action Lawsuits & Class Action Settlements:
- Class actions allege phenylephrine in cold, allergy drugs ineffective
- Recall initiated for Brexafemme antifungal tablets due to possible cross contamination
- Eli Lilly files 10 lawsuits over diabetes drug counterfeits, compounds
- Companies face lawsuits over drug products; FDA approves OTC Narcan, infant RSV vaccine














59 thoughts onTylenol, Mucinex buyer files class action lawsuit over allegedly ineffective ingredient
Yes, and I always wondered why it never seemed to work for me.
I have purchased in the past and recently purchased these items. Please add me.
Me 2 plz!
Please add me
Please add me!
Please add me
Have several boxes. Very concerned.
Add me please
Please add me
I’m taking this now. Have a box in cupboard
I’ve been purchasing Tylenol cold medicine and mucinex over 10 years please contact me regarding this please.
Add me please
Add me