Top Class Actions’s website and social media posts use affiliate links. If you make a purchase using such links, we may receive a commission, but it will not result in any additional charges to you. Please review our Affiliate Link Disclosure for more information.
Vaginal Mesh Lawsuit Update: Ethicon Must Disclose FDA Communications
By Andrea Gressman
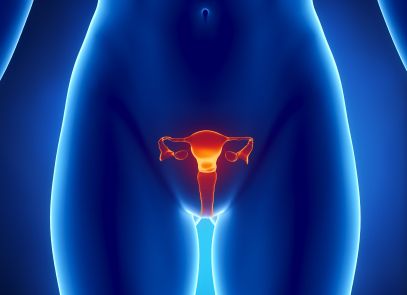
FDA Evaluates Safety of Vaginal Mesh Implants
In 2011, a number of complaints and post-surgery complications from patients using vaginal mesh implants caused the FDA to re-evaluate whether the material was superior to non-mesh options. As the agency took a second look at the risk factors, it issued warnings to the medical community and patients that vaginal mesh implants do not come without risk. Following up these warnings, in January 2012 the FDA ordered more than 30 vaginal mesh implant manufacturers to provide up to three years of data on the safety and effectiveness of their devices.
Between January 2012 and June 2012, Ethicon was an active seller of vaginal mesh implants. As a result, it was in regular communication with the FDA to fulfill the post-market study mandate.
On June 4, 2012, Ethicon said it was going to stop selling four of its vaginal mesh implant systems: Gynecare TCT Secur, Gynecare Prosima, Gynecare Prolift, and Gynecare Proflift+M. The company then asked the FDA to put its post-market study orders on hold.
However, at the time Ethicon asked the FDA to halt its study orders, the company had multiple post-market studies underway. These studies covered the safety and potential complications of using its vaginal mesh implants in detail.
Prior to the August 8 ruling, both Ethicon and its parent company, Johnson & Johnson, were involved in hundreds of vaginal mesh implant lawsuits, but neither entity had submitted paperwork regarding the post-market studies to the New Jersey State Court. Their reasoning was that since Ethicon no longer sold vaginal mesh implants, the data was not relevant.
Vaginal mesh implant lawyers representing Plaintiffs in the Mass Tort requested the data be turned over to help their clients’ cases. Even though Ethicon was no longer actively involved in making or selling vaginal mesh implants, the company was the manufacturer of record for the implants in the ongoing vaginal mesh lawsuit filings.
It is hoped that these new disclosure requirements for Ethicon will provide more information about the nature and type of injuries caused by its Gynecare vaginal mesh implants. This will help evaluate injury claims by the vaginal mesh lawsuit Plaintiffs in New Jersey, and be relevant in the vaginal mesh lawsuit MDL being held in the U.S. District Court for the Southern District of West Virginia.
If you or a loved one has suffered injury or health complications from a vaginal mesh implant, transvaginal mesh impalnt or bladder sling implant, don’t stay silent. Reach out and visit the Transvaginal Mesh, Vaginal Sling, Vaginal Mesh and Bladder Sling Class Action Lawsuit Investigation right away. Provide your information and a team of vaginal mesh implant attorneys will help you find out what compensation you may be entitled to receive as a result of your experience.
Updated August 31st, 2012
{loadposition content_postads}
All medical device, dangerous drug and medical class action and lawsuit news updates are listed in the Drug and Medical Device section of Top Class Actions.
Top Class Actions Legal Statement