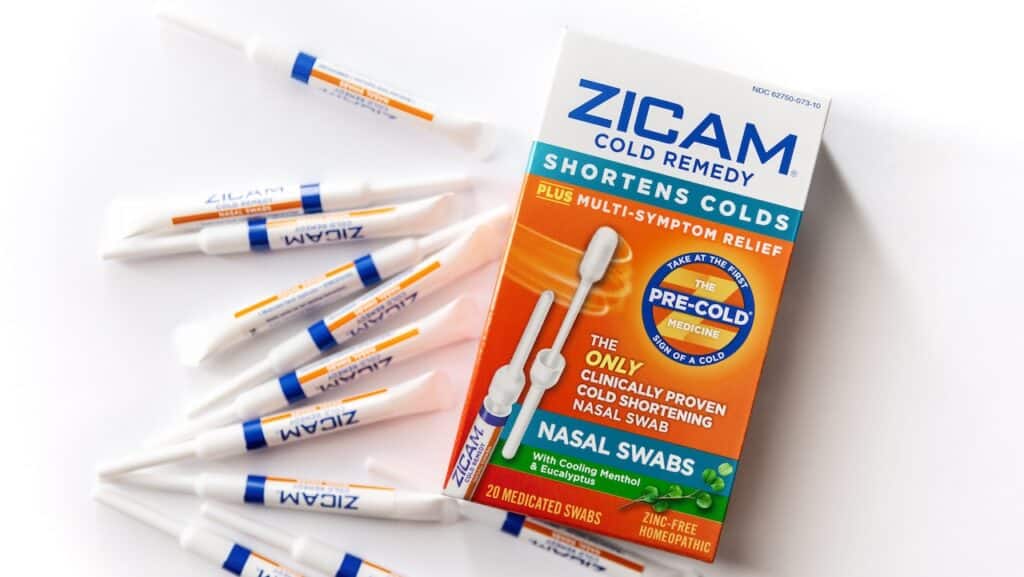
Church & Dwight Zicam recall overview:
- Who: Church & Dwight Co. Inc. is recalling all lots of Zicam Cold Remedy Nasal Swabs, Zicam Nasal AllClear Swabs and Orajel Baby Teething Swabs.
- Why: The recall is due to potential microbial contamination, specifically fungi, in the cotton swab components.
- Where: The recall is effective nationwide in the United States and Puerto Rico.
Church & Dwight Co. is voluntarily recalling several swab products, including Zicam Cold Remedy Nasal Swabs, Zicam Nasal AllClear Swabs and Orajel Baby Teething Swabs, due to the risk of microbial contamination.
The contamination involves fungi in the cotton swab components, posing a significant health risk, especially to children and individuals with compromised immune systems.
The recall, announced on June 6, affects all lots of the specified products within their expiry dates.
The Zicam Cold Remedy Nasal Swabs, identified by UPC 732216301205, are marketed as a zinc-free homeopathic remedy to shorten the duration of colds. The Zicam Nasal AllClear Swabs, with UPC 732216301656, were designed for nasal cleansing but were discontinued in December 2024. Orajel Baby Teething Swabs, carrying UPC 310310400002, are pre-moistened to alleviate teething discomfort in infants and toddlers.
According to the recall notice, “Swabs found to contain microbial contamination can potentially present a significant risk to the health and safety of consumers, including serious and life-threatening blood infections in users whose nasal mucosa may be compromised due to inflammation and mechanical injuries.”
Consumers urged to stop using recalled swabs immediately
The recall notice advises consumers to cease using the affected products immediately and visit www.churchdwightrecall.com for a full refund.
The company has provided a dedicated Consumer Relations team, available at (800) 981-4710, to handle inquiries and process refunds. Additionally, any adverse reactions or quality issues can be reported to the U.S. Food and Drug Administration’s MedWatch Adverse Event Reporting program.
Church & Dwight Co. is taking proactive measures to address the contamination issue by recalling the affected swabs from the market.
The company assures consumers that all other Zicam and Orajel products, including Zicam RapidMelts, remain unaffected by the recall.
Church & Dwight Co. reports that no illnesses or injuries have been linked to the recalled products so far. The company is not currently facing legal action over the recall, but Top Class Actions follows recalls closely as they sometimes lead to class action lawsuits.
Earlier this year, Church & Dwight Co. agreed to a $6 million class action settlement after allegations that its Zicam cold remedies were falsely advertised as able to shorten the duration of colds.
Are you affected by this Church & Dwight Zicam recall? Let us know in the comments.
Don’t Miss Out!
Check out our list of Class Action Lawsuits and Class Action Settlements you may qualify to join!
Read About More Class Action Lawsuits & Class Action Settlements:
- Frito-Lay class action alleges SunChips falsely advertised as 100% whole grain
- Class action claims Ritual Zero Proof adds ‘junk fees’ to online orders
- Class action claims BofA pocketed interest meant for Calif. EDD debit card recipients
- Class action alleges Fox Nation video subscription violates auto-renewal law















114 thoughts onZicam and Orajel swabs recalled over contamination concerns
Unaware of the nasal swab recall , I used the swabs on March 5th of this year , 2026. By early that evening, my lower lip felt odd and looked as though it was swelling. The swelling continued and it was odd. Almost not like. It was identified as an angioedema and I was given Citrizine that reduced the swelling in my lower lip. Although the swelling was reduced, my lip still felt odd as well as my top lip and my gums were swollen. I went to bed as usual that night. When I awoke on the morning of March 6th I did not feel well and swallowing was very painful. The pain increased throughout the day as did my discomfort. I did see an ER doctor on the evening of the 7th. He dismissed my Zicam suggestion and not politely. He said I have a sinus infection. By today, March 8th, 2026, I am literally scared because I am not being treated for what I am convinced is an infection from the fungi contamination in the Zicam Swab. Other symptoms I’m experiencing include: relentless headache, fever, nausea, vomiting, dizziness, diarrhea, difficulty urinating, stomach cramps that intensify if I eat or drink, absence of taste and appetite, sore white throat, body aches and weakness. I truly believe I could be Zicams first case to become infected by the Fungal contamination. Making it all too coincidental, every number on the box recalled Lot #/barcode all match mine perfectly. I’m really reaching out because I’m sick, really sick and cannot get any medical personnel to take me seriously.
I still have some of the zicam. And my husband and children and myself used them. Please add me to the list!
I have no receipts, but my husband and I have used Zicam in the past and recently. Please put me on your list.