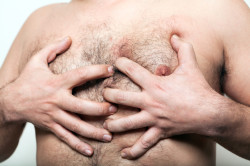
The psychiatric medication Risperdal can be linked to abnormal growth of breast tissue in men, which in some more refractory cases can require surgery to correct.
Risperdal, also known under its generic name risperidone, was originally approved by the FDA in 1993. Today, Risperdal is indicated for the treatment of schizophrenia, the manic episodes of bipolar disorder, and irritability associated with autistic disorder.
How Risperdal works is not known for certain, but it may work by suppressing the action of dopamine Type 2 (or D2) and serotonin Type 2 (or 5HT2) receptors. But like other drugs that antagonize D2 receptors, Risperdal increases the presence of the hormone prolactin. Prolactin is better known for stimulating breast growth and milk production in women, but it can have the same effect in men.
Surgery for Risperdal Gynecomastia
Gynecomastia usually resolves after its root cause is successfully dealt with. But in rare cases gynecomastia persists, and in those more stubborn cases, male breast reduction surgery may be necessary.
Male breast reduction surgery typically addresses both the excess breast tissue and accompanying increase in fat that both occur in gynecomastia. The operation usually starts with liposuction to remove the newly built-up fat. Then, to get to the breast tissue itself, the surgeon makes a semicircular incision along the bottom of the areola. The surgeon then excises the excess breast tissue through that incision.
To wrap up the surgery, the surgeon must place drains in the liposuction incisions that must be left in place for two days. The patient must also wear a compression garment around the chest that promotes healing. After seven to 10 days, the sutures can be removed. The patient will have to avoid any strenuous exertion for about three weeks, but most patients can resume sedentary work after five days.
Risperdal Lawsuits
Risperdal gynecomastia can be emotionally taxing for men, and even more so for boys. Many of the Risperdal lawsuits now pending are being brought by men who were prescribed Risperdal when they were boys or adolescents and who later developed Risperdal gynecomastia.
The use of Risperdal in these young boys was allegedly brought about in part through Johnson & Johnson’s own marketing efforts. In 2013, Johnson & Johnson agreed to pay a record-setting $2.2 billion penalty to settle both criminal and civil allegations, in part for unlawfully marketing Risperdal for use in children at a time when the FDA had approved the drug only for use in adults. (While individual doctors may prescribe a drug for any use they see fit, it’s unlawful for a drug’s manufacturer to promote the drug for any use other than those approved by the FDA.)
In a later Risperdal lawsuit, a former FDA regulator testified that Johnson and Johnson knew as early as 2001 about the link between Risperdal and gynecomastia. Plaintiffs in several Risperdal lawsuits allege the company purposely withheld information about that link from patients and their doctors.
Today there are over 1,300 such Risperdal lawsuits pending against Johnson & Johnson. Results of recent bellwether trials in Pennsylvania have been mixed. In the first such trial, the jury awarded the plaintiff $2.5 million. But the second such trial resulted in a verdict for the defendants. The jury there nevertheless found that Johnson & Johnson failed to adequately warn patients and doctors about the risk of Risperdal gynecomastia.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The Risperdal attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or Risperdal class action lawsuit is best for you. [In general, Risperdal lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.
Get Help – It’s Free
Join a Free Risperdal Class Action Lawsuit Investigation
If you or your son took Risperdal between the ages of 10 and 18 years old and suffered gynecomastia (male breast growth), male breast pain, nipple pain, or nipple discharge, you may be entitled to compensation. See if you qualify by submitting your information below for a free and confidential case review.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Oops! We could not locate your form.












