 Eliquis is one of a new generation of anticoagulants that the FDA began approving in 2010.
Eliquis is one of a new generation of anticoagulants that the FDA began approving in 2010.
These new drugs were designed to offer certain advantages over warfarin, which had been the go-to anticoagulant since the 1950s.
In particular, these new drugs were designed to be administered without the periodic blood testing and dosage adjustment that may be necessary with warfarin.
However, at the time each of these drugs was released, there was no antidote (or “reversal agent”) that could reverse their anticoagulant effect in the event of a brain bleed or other internal bleeding.
If a patient suffered a brain bleed or needed emergency surgery while on these drugs, they would simply have to discontinue the drug and wait for the body to finish metabolizing it. This fact allowed warfarin to retain at least one advantage over the new drugs, since its effect can be readily reversed by administering vitamin K.
Anticoagulants like Eliquis are often prescribed for patients with atrial fibrillation. These patients are at risk for developing blood clots in the heart that could travel to the brain and cause a stroke. Anticoagulants reduce this risk of stroke by preventing these clots from forming.
The down side of this anticoagulant action is that it can lead to excessive bleeding. By reducing the blood’s ability to clot, anticoagulants like Eliquis decrease the body’s ability to keep bleeding under control.
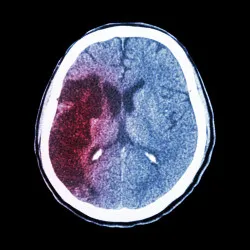
The anticoagulant effect of Eliquis may be linked to an increased risk of bleeding on the brain, a condition sometimes referred to as hemorrhagic stroke. This type of brain bleed is different from the kind of stoke Eliquis is meant to prevent, known as an ischemic stroke.
A brain bleed results not from a clot blocking blood flow to the brain but from damage to a blood vessel that results in bleeding within or on the outside of the brain.
Despite the dangers of bleeding on the brain and other types of excessive bleeding, Eliquis was introduced to the market without any reversal agent, leaving doctors without that effective option for stopping a brain bleed once it starts. An antidote for Eliquis, andexanet alfa, has been developed and is currently awaiting FDA approval, but it has not yet been made available for patients.
Concerns have been raised about some of the clinical trials used to provide data on which the FDA based its approval of Eliquis. Evidence of impropriety at a clinical trial site in China has raised serious questions as to the reliability of the trial results.
One manager of a trial site was fired for altering records to hide violations of clinical practice guidelines. The FDA found inconsistencies throughout the trial data. Some adverse events were reported late or not at all.
Eliquis Brain Bleed Lawsuits
The first known Eliquis lawsuit was filed in July of 2015 by the widow of a man who died of a brain bleed and gastrointestinal hemorrhage while taking Eliquis. The plaintiff argues that the manufacturers of Eliquis purposely concealed information about the risks of bleeding from the patient and his doctor.
Since thousands of earlier lawsuits were filed over other new anticoagulant medications, a similar wave of Eliquis lawsuits may be on the way.
Do YOU have a legal claim? Fill out the form on this page now for a free, immediate, and confidential case evaluation. The Eliquis attorneys who work with Top Class Actions will contact you if you qualify to let you know if an individual lawsuit or Eliquis class action lawsuit is best for you. [In general, Eliquis lawsuits are filed individually by each plaintiff and are not class actions.] Hurry — statutes of limitations may apply.
Get Help – It’s Free
Join a Free Eliquis Class Action Lawsuit Investigation
If you or a loved one took Eliquis (apixaban) and suffered injuries such as uncontrollable internal bleeding, gastrointestinal bleeding, hemorrhaging, kidney bleeding or death, you may have a legal claim. See if you qualify by filling out the short form below.
An attorney will contact you if you qualify to discuss the details of your potential case at no charge to you.
Oops! We could not locate your form.
ATTORNEY ADVERTISING
Top Class Actions is a Proud Member of the American Bar Association
LEGAL INFORMATION IS NOT LEGAL ADVICE
Top Class Actions Legal Statement
©2008 – 2026 Top Class Actions® LLC
Various Trademarks held by their respective owners
This website is not intended for viewing or usage by European Union citizens.













Just had an attorney friend die of brain bleed
My wife was taking Eliquis for about 3 weeks when her brain hemorrhaged.